Write The Electronic Configuration Of Sodium
News Leon
Apr 05, 2025 · 6 min read
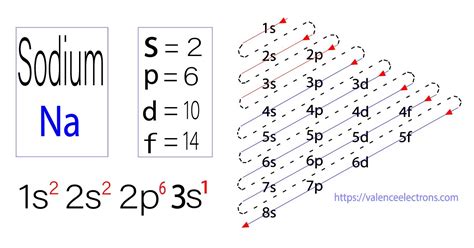
Table of Contents
Unveiling the Secrets of Sodium's Electronic Configuration: A Deep Dive
Sodium, a ubiquitous element found in table salt and crucial for biological processes, possesses a fascinating electronic configuration that dictates its chemical behavior and properties. Understanding this configuration is key to grasping its reactivity and role in various chemical reactions. This comprehensive guide will delve into the electronic configuration of sodium, exploring the underlying principles, its implications, and its significance in chemistry.
Understanding Electronic Configuration: The Foundation
Before we dive into the specifics of sodium's configuration, let's establish a foundational understanding of what electronic configuration actually means. Simply put, an electronic configuration describes the arrangement of electrons within the electron shells and subshells of an atom. This arrangement follows specific rules governed by quantum mechanics, ensuring stability and influencing the atom's interactions with other atoms.
The Quantum Mechanical Model: The Rules of the Game
The electronic configuration is determined by the principles of quantum mechanics. Key concepts include:
-
Principal Quantum Number (n): This number defines the energy level or shell of an electron. It can be any positive integer (1, 2, 3, etc.). Higher n values indicate higher energy levels and greater distance from the nucleus.
-
Azimuthal Quantum Number (l): This number describes the subshell within a principal shell. It ranges from 0 to n-1. Subshells are often denoted by letters: l=0 (s), l=1 (p), l=2 (d), l=3 (f).
-
Magnetic Quantum Number (ml): This defines the specific orbital within a subshell. It ranges from -l to +l, including 0. Each orbital can hold a maximum of two electrons.
-
Spin Quantum Number (ms): This describes the intrinsic angular momentum of an electron, often represented as +1/2 (spin up) or -1/2 (spin down). The Pauli Exclusion Principle states that no two electrons in an atom can have the same set of four quantum numbers.
These quantum numbers dictate the allowed energy levels and the spatial distribution of electrons within an atom.
Determining Sodium's Electronic Configuration: A Step-by-Step Approach
Sodium (Na) has an atomic number of 11, meaning it has 11 protons and 11 electrons in a neutral atom. To determine its electronic configuration, we follow the Aufbau principle, Hund's rule, and the Pauli exclusion principle.
The Aufbau Principle: Filling the Shells
The Aufbau principle states that electrons fill the lowest energy levels first. The order of filling is generally: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on. However, there are exceptions to this rule, especially for transition metals.
Hund's Rule: Maximizing Spin Multiplicity
Hund's rule states that electrons will individually occupy each orbital within a subshell before pairing up. This maximizes the total spin multiplicity, leading to greater stability.
The Pauli Exclusion Principle: One Electron Per Orbital
The Pauli exclusion principle dictates that no two electrons in an atom can have the same four quantum numbers. This means each orbital can hold a maximum of two electrons, with opposite spins.
Applying the Rules to Sodium (Na, Z=11)
-
1s²: The first shell (n=1) has only one subshell (s), which can hold a maximum of two electrons. Both electrons fill this lowest energy level.
-
2s²: The second shell (n=2) has an s subshell, which also holds two electrons. These two electrons fill this subshell.
-
2p⁶: The second shell also has a p subshell, which can hold up to six electrons (three orbitals, each holding two electrons). All six electrons fill this subshell.
-
3s¹: After filling the 2p subshell, we move to the third shell (n=3). The 3s subshell receives the remaining electron.
Therefore, the complete electronic configuration of sodium is 1s²2s²2p⁶3s¹. This can also be represented in a shorthand notation using the noble gas configuration of neon (Ne), which is [Ne]3s¹.
Significance of Sodium's Electronic Configuration: Implications for Reactivity
Sodium's electronic configuration is crucial in understanding its chemical properties and reactivity. The single electron in the 3s orbital is relatively loosely held by the nucleus. This makes it easy to lose this electron to achieve a stable octet configuration, similar to the noble gas neon.
Ionization Energy and Reactivity
The ionization energy is the energy required to remove an electron from an atom. Due to the loosely held 3s electron, sodium has a relatively low ionization energy. This means it readily loses its valence electron to form a positively charged ion, Na⁺. This tendency to lose an electron and form a cation is a hallmark of alkali metals, the group to which sodium belongs.
Chemical Bonding and Compound Formation
Sodium's tendency to lose an electron facilitates its participation in ionic bonding. It readily reacts with electronegative elements, like chlorine (Cl), transferring its valence electron to form an ionic compound, sodium chloride (NaCl) or table salt. In this compound, sodium exists as a Na⁺ cation and chlorine as a Cl⁻ anion, held together by electrostatic forces.
Sodium's Role in Biology and Industry
Sodium's unique properties, stemming directly from its electronic configuration, make it indispensable in various biological and industrial applications:
Biological Significance: Essential for Life
Sodium ions (Na⁺) play a crucial role in various biological processes:
-
Nerve Impulse Transmission: Sodium ions are vital for the transmission of nerve impulses. The movement of Na⁺ ions across cell membranes generates electrical signals that enable communication within the nervous system.
-
Muscle Contraction: Similar to nerve impulse transmission, Na⁺ ions are essential for muscle contraction, facilitating the interaction of muscle proteins and enabling movement.
-
Fluid Balance: Sodium ions help regulate fluid balance in the body, maintaining the proper osmotic pressure within cells and tissues.
Industrial Applications: A Versatile Element
Sodium's versatility finds applications in various industries:
-
Sodium Chloride (NaCl): Widely used as table salt, it's also employed in food preservation, de-icing roads, and various industrial processes.
-
Sodium Hydroxide (NaOH): A strong alkali, commonly known as lye or caustic soda, it's used in soap making, paper production, and numerous chemical processes.
-
Sodium Lamps: Sodium vapor lamps provide efficient and bright illumination, frequently used in street lighting and other outdoor applications.
-
Sodium-Sulfur Batteries: These high-energy-density batteries are used in various applications requiring high power output and long lifespan.
Conclusion: A Fundamental Understanding
The electronic configuration of sodium, 1s²2s²2p⁶3s¹, is not merely a theoretical concept; it's the fundamental key to understanding its chemical behavior, reactivity, and biological significance. The ease with which sodium loses its valence electron to achieve a stable octet configuration dictates its participation in ionic bonding, its low ionization energy, and its crucial role in biological processes. From our table salt to nerve impulses and industrial applications, sodium's impact is undeniable, all stemming from the arrangement of its 11 electrons. Understanding this simple yet profound configuration allows us to appreciate the remarkable properties and importance of this everyday element.
Latest Posts
Latest Posts
-
Difference Between Cls And Self In Python
Apr 05, 2025
-
The Cell Wall In Bacteria Is Primarily Composed Of
Apr 05, 2025
-
What Is 2 3 Repeating As A Fraction
Apr 05, 2025
-
Constant Returns To Scale Production Function
Apr 05, 2025
-
What Is The Index Of Refraction For Crown Glass
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Write The Electronic Configuration Of Sodium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
