Which Of The Following Is Chemically Inert Unreactive
News Leon
Apr 02, 2025 · 5 min read
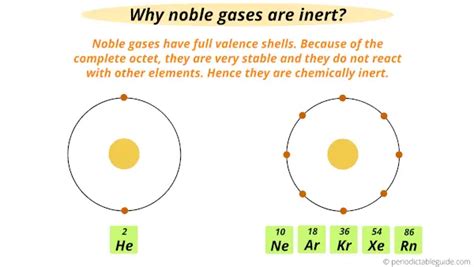
Table of Contents
Which of the Following is Chemically Inert/Unreactive? Understanding Chemical Inertness
The question, "Which of the following is chemically inert/unreactive?" hinges on understanding chemical reactivity. Chemical inertness, or lack of reactivity, signifies a substance's resistance to undergoing chemical changes or reactions under normal conditions. Many factors influence a substance's reactivity, including its electronic configuration, bond strength, and the presence of catalysts. This article delves deep into the concept of chemical inertness, examining various elements and compounds, and clarifying which are generally considered unreactive.
Defining Chemical Inertness: More Than Just "Doesn't React"
While the simplest definition of chemical inertness is "doesn't react," the reality is more nuanced. A substance deemed inert under one set of conditions might react under different, more extreme conditions (high temperature, pressure, presence of a catalyst, etc.). True chemical inertness implies a significant resistance to reaction across a broad range of conditions, meaning it requires substantial energy input to initiate a reaction.
Several factors contribute to a substance's inertness:
-
Full Valence Shells: Atoms strive to achieve a stable electron configuration, usually a full valence shell (outer electron shell). Elements with full valence shells, like noble gases, generally exhibit high inertness because they don't readily gain, lose, or share electrons.
-
Strong Bonds: Substances with exceptionally strong bonds require a large amount of energy to break those bonds, thereby reducing their reactivity. For example, many diatomic molecules with triple bonds (like nitrogen, N₂) exhibit relatively high inertness.
-
High Ionization Energy: High ionization energy means it takes a substantial amount of energy to remove an electron from an atom. This makes it less likely to participate in reactions involving electron donation.
-
High Electronegativity: High electronegativity indicates an atom's strong attraction for electrons. This can prevent it from readily sharing or losing electrons, hindering its reactivity.
-
Kinetic Factors: Even if thermodynamically favorable, a reaction might not occur at an appreciable rate due to slow reaction kinetics. This often relates to high activation energy which is the energy barrier to initiation of the reaction.
Examining Commonly Considered Inert Substances
Let's examine some substances frequently associated with chemical inertness:
1. Noble Gases (Group 18): The Paragon of Inertness
The noble gases (Helium, Neon, Argon, Krypton, Xenon, Radon) are the quintessential examples of chemically inert elements. Their filled valence shells (eight electrons, except for helium with two) provide exceptional stability. They rarely participate in chemical reactions, earning them their name.
-
Helium (He): The lightest noble gas, incredibly unreactive. Used in balloons and cryogenics.
-
Neon (Ne): Known for its bright red-orange glow in neon signs, extremely unreactive.
-
Argon (Ar): The most abundant noble gas in the atmosphere; used in welding and light bulbs.
-
Krypton (Kr), Xenon (Xe), Radon (Rn): While generally inert, these heavier noble gases can participate in reactions under specific extreme conditions (e.g., high pressure, presence of highly reactive species). However, their reactivity remains extremely low compared to other elements.
2. Nitrogen (N₂): A Reluctant Reactor
Nitrogen gas, composing about 78% of Earth's atmosphere, is a diatomic molecule (N₂) with a strong triple bond. This strong bond requires a considerable amount of energy to break, rendering it relatively inert under normal conditions. Although nitrogen is essential for life and participates in numerous biological processes, its inherent inertness necessitates specialized biological nitrogen fixation mechanisms. At high temperatures and pressures, or with a catalyst, nitrogen can react. The Haber-Bosch process, for instance, uses high temperature, pressure and an iron catalyst to convert N₂ and H₂ into ammonia (NH₃).
3. Diamond: A Crystalline Inertness
Diamond, an allotrope of carbon, boasts a highly stable crystal lattice structure. Each carbon atom forms strong covalent bonds with four other carbon atoms, creating a rigid, inert structure. Although diamond can be burned in oxygen at high temperatures, it's remarkably resistant to most chemical reactions under normal conditions.
4. Certain Fluorocarbons: Engineered Inertness
Some fluorocarbons, compounds containing carbon and fluorine, exhibit significant chemical inertness. Their strong carbon-fluorine bonds contribute to their stability and resistance to reactions. These materials find applications in non-stick cookware (Teflon) and refrigerants (though many such refrigerants have been phased out due to environmental concerns). The extreme strength of the C-F bond contributes to their inert nature.
5. Gold (Au): The Noble Metal
Gold is a classic example of a noble metal, meaning it exhibits high resistance to oxidation and corrosion. Its electronic configuration and relatively high electronegativity contribute to its inertness. While gold can react with certain highly reactive substances (like aqua regia, a mixture of nitric and hydrochloric acids), it remains remarkably unreactive under typical environmental conditions.
Factors Affecting Apparent Inertness: The Context Matters
It's crucial to emphasize that the term "inert" is relative and context-dependent. Several factors can influence a substance's apparent reactivity:
-
Temperature: Higher temperatures generally increase reaction rates, potentially overcoming the energy barrier for even seemingly inert substances.
-
Pressure: Increased pressure can favor reactions that involve a decrease in the number of gas molecules.
-
Catalysts: Catalysts can dramatically lower the activation energy, accelerating reactions that would otherwise be extremely slow.
-
Presence of other reactants: Highly reactive substances can induce reactions in otherwise inert materials.
-
Surface area: A larger surface area increases the contact between reactants, increasing the reaction rate.
Conclusion: Understanding the Nuances of Chemical Inertness
While certain elements and compounds are commonly referred to as "chemically inert," it's essential to appreciate that this term is a relative descriptor. Truly inert substances exhibit remarkable resistance to chemical reactions under a wide range of conditions due to factors such as full valence shells, strong bonds, and high ionization energies. However, even the most inert substances can react under extreme conditions or with the right catalysts. This understanding is critical across various fields, from materials science and chemical engineering to biology and environmental science. Understanding the factors that influence chemical reactivity allows us to predict and manipulate chemical processes with greater precision and control. The identification of a chemically inert substance always depends on the specific conditions under consideration.
Latest Posts
Latest Posts
-
Oxidation Number Of Carbon In Co
Apr 03, 2025
-
Is Friction A Non Conservative Force
Apr 03, 2025
-
Which Of The Following Graphs Represents A One To One Function
Apr 03, 2025
-
Is B2 2 Paramagnetic Or Diamagnetic
Apr 03, 2025
-
Capital Of Karnataka State In India
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is Chemically Inert Unreactive . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
