Is B2 2- Paramagnetic Or Diamagnetic
News Leon
Apr 03, 2025 · 5 min read
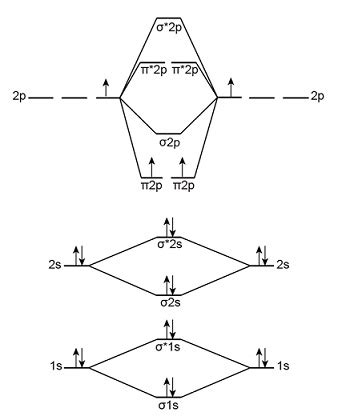
Table of Contents
Is B2 2- Paramagnetic or Diamagnetic? Delving into Molecular Orbital Theory
Determining the magnetic properties of a molecule like B₂²⁻ requires a solid understanding of molecular orbital (MO) theory. This article will delve into the intricacies of MO theory, specifically applying it to the B₂²⁻ anion, to definitively answer whether it's paramagnetic or diamagnetic. We'll explore the electronic configuration, bond order, and the implications of unpaired electrons. Furthermore, we'll examine the broader context of boron's behavior in different chemical environments.
Understanding Paramagnetism and Diamagnetism
Before diving into the specifics of B₂²⁻, let's establish the fundamental difference between paramagnetism and diamagnetism:
-
Diamagnetism: This is a fundamental property of all matter. Diamagnetic substances possess paired electrons, meaning all electron spins are coupled. When exposed to an external magnetic field, they generate a weak, opposing magnetic field. They are weakly repelled by magnets.
-
Paramagnetism: This property arises from the presence of unpaired electrons. These unpaired electrons possess individual magnetic moments that align with an external magnetic field, leading to a net attraction. Paramagnetic substances are weakly attracted to magnets.
Molecular Orbital Theory: The Key to Understanding B₂²⁻
Molecular orbital (MO) theory provides a powerful framework for predicting the magnetic properties of molecules. Unlike valence bond theory, which focuses on individual atomic orbitals forming bonds, MO theory considers the combination of atomic orbitals to form molecular orbitals that encompass the entire molecule.
These molecular orbitals can be categorized as bonding (lower energy, stabilizing) and antibonding (higher energy, destabilizing). Electrons fill these molecular orbitals according to the Aufbau principle (lowest energy levels first) and Hund's rule (maximum multiplicity—maximizing unpaired electrons).
Constructing the Molecular Orbital Diagram for B₂²⁻
Boron (B) has an atomic number of 5, with an electronic configuration of 1s²2s²2p¹. In the formation of B₂²⁻, we consider the valence electrons (2s and 2p orbitals). The diatomic molecule B₂ initially involves two boron atoms contributing a total of six valence electrons (two 2s and four 2p electrons). However, we're dealing with B₂²⁻, which means it has gained two additional electrons, bringing the total electron count to eight.
The molecular orbital diagram for B₂²⁻ is constructed as follows:
-
σ₂s and σ₂s:* The 2s atomic orbitals combine to form a bonding σ₂s and an antibonding σ₂s* molecular orbital.
-
σ₂p, π₂p, π₂p, and σ₂p:** The 2p atomic orbitals combine to form one bonding σ₂p, two degenerate bonding π₂p orbitals, two degenerate antibonding π₂p* orbitals, and one antibonding σ₂p* orbital.
The order of energy levels can vary depending on the specific molecule, but a common ordering (and the one we'll use for B₂²⁻) is: σ₂s < σ₂s* < σ₂p < π₂p < π₂p* < σ₂p*.
Filling the Molecular Orbitals for B₂²⁻
With eight valence electrons in B₂²⁻, we fill the molecular orbitals according to the Aufbau principle and Hund's rule. This leads to the following electronic configuration:
(σ₂s)² (σ₂s*)² (σ₂p)² (π₂p)⁴
Notice that all electrons are paired.
Determining the Magnetic Property of B₂²⁻
Crucially, the electronic configuration shows that all electrons in B₂²⁻ are paired. The presence of only paired electrons signifies that B₂²⁻ is diamagnetic. It will exhibit a weak repulsion in the presence of an external magnetic field.
Bond Order and its Relation to Stability
The bond order is a useful concept to determine the strength and stability of a chemical bond. It's calculated as half the difference between the number of electrons in bonding molecular orbitals and the number of electrons in antibonding molecular orbitals:
Bond Order = (Number of bonding electrons - Number of antibonding electrons) / 2
For B₂²⁻:
Bond Order = (6 - 2) / 2 = 2
A bond order of 2 indicates a stable double bond. This further supports the stability of the B₂²⁻ anion.
Comparing B₂²⁻ to other Boron Species
It's instructive to compare B₂²⁻ to other boron-containing species to understand the influence of electron count and charge on magnetic properties:
-
B₂: With only six valence electrons, B₂ would have the configuration (σ₂s)² (σ₂s*)² (σ₂p)² . This results in two unpaired electrons in the π₂p orbitals, making it paramagnetic. Its bond order is 1.
-
B₂⁺: Losing an electron compared to B₂ would result in one unpaired electron, also rendering it paramagnetic.
-
B₂⁻: Gaining an electron compared to B₂ would lead to a single unpaired electron in the π₂p orbitals, rendering it paramagnetic.
This comparison highlights the significant role of electron count in dictating the magnetic properties of these boron-containing species. The additional electrons in B₂²⁻ completely fill the bonding and antibonding orbitals, leading to diamagnetism.
Experimental Verification and Computational Chemistry
While MO theory provides a powerful predictive tool, experimental verification is crucial. Techniques like electron paramagnetic resonance (EPR) spectroscopy are capable of detecting unpaired electrons. The absence of an EPR signal for B₂²⁻ would further support its diamagnetic nature. Furthermore, computational chemistry methods, like density functional theory (DFT) calculations, can be employed to simulate the electronic structure and predict the magnetic properties of B₂²⁻, providing an independent confirmation of the MO theory predictions. These methods often include sophisticated approaches to determine electron correlation and accurately capture the energy differences between orbitals.
Conclusion: B₂²⁻ is Diamagnetic
In conclusion, based on the molecular orbital diagram, electronic configuration, and the absence of unpaired electrons, the B₂²⁻ anion is definitively diamagnetic. Its diamagnetism stems from the complete pairing of all valence electrons within the molecular orbitals. This is a direct consequence of adding two extra electrons to the neutral B₂ molecule, filling all available bonding and lower-lying antibonding molecular orbitals. Understanding this requires a firm grasp of MO theory and the interplay between bonding and antibonding orbitals. Further support for this conclusion could be obtained from experimental methods and computational simulations. The stability is confirmed by the bond order calculation, giving a robust double bond. The comparison with related species provides a wider perspective on the influence of charge and electron count on magnetic behavior in boron-containing molecules.
Latest Posts
Latest Posts
-
Is A Euglena A Prokaryote Or Eukaryote
Apr 04, 2025
-
Containing Two Different Alleles For A Trait
Apr 04, 2025
-
Which Of The Following Is A Non Renewable Source Of Energy
Apr 04, 2025
-
What Binds To The Exposed Cross Bridges On Actin
Apr 04, 2025
-
All Squares Are Rectangles And Rhombuses
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Is B2 2- Paramagnetic Or Diamagnetic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
