The Mass Of A Mole Of Nacl Is The
News Leon
Apr 03, 2025 · 4 min read
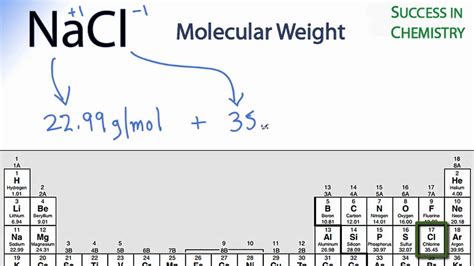
Table of Contents
The Mass of a Mole of NaCl: Unpacking Avogadro's Number and Molar Mass
The seemingly simple question, "What is the mass of a mole of NaCl?" opens a door to a fundamental concept in chemistry: the mole. Understanding this concept is crucial for anyone working with chemical reactions, stoichiometry, and quantitative analysis. This article delves deep into the calculation, explaining the underlying principles and offering practical examples to solidify your understanding.
Understanding the Mole: The Chemist's Counting Unit
Before tackling the mass of a mole of NaCl, we need to grasp the concept of the mole itself. A mole is not a unit of weight or volume; it's a unit of amount of substance. Just as a dozen represents 12 items, a mole represents Avogadro's number (approximately 6.022 x 10<sup>23</sup>) of entities. These entities can be atoms, molecules, ions, or even formula units, depending on the context.
Avogadro's number is a fundamental constant in chemistry, linking the microscopic world of atoms and molecules to the macroscopic world of grams and moles. It acts as a conversion factor between the number of particles and the mass of those particles.
Molar Mass: The Bridge Between Grams and Moles
The molar mass of a substance is the mass of one mole of that substance, expressed in grams per mole (g/mol). It's numerically equal to the atomic or molecular weight of the substance, but with the units changed from atomic mass units (amu) to grams. This equivalence is a direct consequence of Avogadro's number.
For elemental substances, the molar mass is simply the atomic weight from the periodic table. For example, the molar mass of carbon (C) is approximately 12.01 g/mol. However, for compounds like NaCl (sodium chloride), the molar mass is the sum of the molar masses of its constituent atoms.
Calculating the Molar Mass of NaCl
Sodium chloride (NaCl) is an ionic compound composed of sodium (Na) and chlorine (Cl) ions. To calculate its molar mass, we need the atomic weights of sodium and chlorine from the periodic table:
- Sodium (Na): Approximately 22.99 g/mol
- Chlorine (Cl): Approximately 35.45 g/mol
Therefore, the molar mass of NaCl is:
22.99 g/mol (Na) + 35.45 g/mol (Cl) = 58.44 g/mol
This means that one mole of NaCl has a mass of approximately 58.44 grams.
The Mass of a Mole of NaCl: Practical Applications
Understanding the mass of a mole of NaCl has numerous practical applications in chemistry:
1. Stoichiometric Calculations:
Stoichiometry involves calculating the quantities of reactants and products in a chemical reaction. Knowing the molar mass of NaCl allows us to convert between grams of NaCl and moles of NaCl, a crucial step in stoichiometric calculations. For example, if we have a reaction involving 10 grams of NaCl, we can convert this to moles using the molar mass:
10 g NaCl × (1 mol NaCl / 58.44 g NaCl) ≈ 0.171 moles NaCl
2. Solution Preparation:
Molar mass is essential for preparing solutions of a specific concentration. For example, to prepare a 1 Molar (1 M) solution of NaCl, we need to dissolve one mole (58.44 g) of NaCl in one liter of water.
3. Titrations:
Titrations are quantitative analytical techniques used to determine the concentration of a solution. The molar mass of NaCl plays a crucial role in calculating the concentration of a solution during a titration involving NaCl.
4. Gravimetric Analysis:
Gravimetric analysis is another quantitative analytical technique that involves measuring the mass of a precipitate. If the precipitate involves NaCl, its molar mass is used in calculating the amount of the analyte present in the original sample.
Beyond the Basics: Isotopes and Variations in Molar Mass
The molar mass values we use are typically weighted averages of the different isotopes of an element. Isotopes are atoms of the same element with different numbers of neutrons. This means that the actual mass of a single NaCl molecule might vary slightly depending on the specific isotopes of sodium and chlorine present. However, the weighted average molar mass provides a highly accurate representation for most practical purposes.
Error Analysis and Precision
The precision of our calculation of the molar mass of NaCl depends on the precision of the atomic weights used. The periodic table typically provides atomic weights to several decimal places, reflecting the natural abundance of various isotopes. The slight variations in reported atomic weights from different sources will lead to minor differences in calculated molar mass. However, these differences are usually insignificant for most chemical applications.
Conclusion: The Mole – A Cornerstone of Chemistry
The mass of a mole of NaCl, approximately 58.44 grams, is not just a number; it's a fundamental concept linking the atomic world to the macroscopic world. Understanding the mole, Avogadro's number, and molar mass is paramount for success in chemistry. This knowledge empowers us to perform accurate stoichiometric calculations, prepare solutions of precise concentrations, and utilize analytical techniques like titrations and gravimetric analysis. The seemingly simple question about the mass of a mole of NaCl opens up a vast landscape of chemical understanding and application. Mastering this concept provides a strong foundation for further explorations in chemistry and related scientific fields. Further study of concepts like molar volume, solution concentration units (e.g., molality, normality), and chemical kinetics will build upon this foundational knowledge.
Latest Posts
Latest Posts
-
Sodium Sulfate Barium Chloride Balanced Equation
Apr 04, 2025
-
Which Of The Following Compounds Has The Lowest Boiling Point
Apr 04, 2025
-
Does Ionization Increase From Left To Right
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about The Mass Of A Mole Of Nacl Is The . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
