Which Of The Following Compounds Has The Lowest Boiling Point
News Leon
Apr 04, 2025 · 5 min read
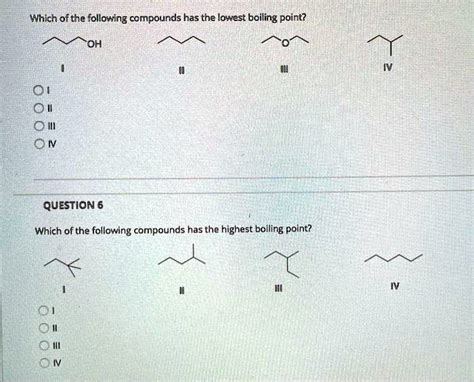
Table of Contents
- Which Of The Following Compounds Has The Lowest Boiling Point
- Table of Contents
- Which of the Following Compounds Has the Lowest Boiling Point? A Deep Dive into Intermolecular Forces
- Understanding Intermolecular Forces
- Factors Affecting Boiling Point
- Comparing Boiling Points: A Case Study
- Advanced Considerations and Exceptions
- Practical Applications and Further Exploration
- Latest Posts
- Latest Posts
- Related Post
Which of the Following Compounds Has the Lowest Boiling Point? A Deep Dive into Intermolecular Forces
Determining which compound possesses the lowest boiling point from a given set requires a thorough understanding of intermolecular forces (IMFs). Boiling point is directly related to the strength of these forces; stronger IMFs lead to higher boiling points because more energy is needed to overcome the attractions between molecules and transition them from the liquid to the gaseous phase. This article will explore the various types of IMFs and provide a step-by-step approach to predicting boiling points, ultimately allowing you to confidently answer the question: which compound boils at the lowest temperature?
Understanding Intermolecular Forces
Before diving into specific examples, let's review the key intermolecular forces:
1. London Dispersion Forces (LDFs): These are the weakest IMFs and are present in all molecules, regardless of their polarity. LDFs arise from temporary, instantaneous dipoles created by the fluctuating electron distribution around atoms. Larger, more polarizable molecules with greater surface area experience stronger LDFs.
2. Dipole-Dipole Forces: These forces exist between polar molecules, meaning molecules with a permanent dipole moment due to a difference in electronegativity between atoms. The positive end of one molecule attracts the negative end of another. Dipole-dipole forces are stronger than LDFs.
3. Hydrogen Bonding: This is a special type of dipole-dipole interaction that occurs when a hydrogen atom is bonded to a highly electronegative atom (N, O, or F). The hydrogen atom develops a significant partial positive charge, strongly attracting the lone pairs of electrons on another electronegative atom in a nearby molecule. Hydrogen bonding is the strongest type of IMF.
Factors Affecting Boiling Point
Several factors influence the boiling point of a compound, all intricately linked to the strength of its IMFs:
-
Molecular Weight: Larger molecules generally have higher boiling points due to stronger LDFs. Increased molecular weight means more electrons, leading to greater polarizability and stronger temporary dipoles.
-
Molecular Shape: A linear molecule generally has a higher boiling point than a branched molecule with the same molecular weight because linear molecules have a larger surface area for interaction, resulting in stronger LDFs.
-
Polarity: Polar molecules have higher boiling points than nonpolar molecules of similar molecular weight due to the presence of dipole-dipole forces in addition to LDFs. The more polar the molecule, the stronger the dipole-dipole forces and the higher the boiling point.
-
Hydrogen Bonding: The presence of hydrogen bonding dramatically increases the boiling point. Compounds capable of hydrogen bonding have significantly higher boiling points compared to those with only LDFs or dipole-dipole forces.
Comparing Boiling Points: A Case Study
Let's consider a hypothetical example to illustrate how to determine which compound has the lowest boiling point:
Scenario: Which of the following compounds has the lowest boiling point: methane (CH₄), ethanol (CH₃CH₂OH), chloromethane (CH₃Cl), and propane (CH₃CH₂CH₃)?
Step 1: Identify the types of Intermolecular Forces:
- Methane (CH₄): Nonpolar, only LDFs.
- Ethanol (CH₃CH₂OH): Polar, capable of hydrogen bonding.
- Chloromethane (CH₃Cl): Polar, dipole-dipole forces and LDFs.
- Propane (CH₃CH₂CH₃): Nonpolar, only LDFs.
Step 2: Compare Molecular Weights:
- Methane (MW = 16 g/mol)
- Ethanol (MW = 46 g/mol)
- Chloromethane (MW = 50.5 g/mol)
- Propane (MW = 44 g/mol)
Step 3: Analyze the Intermolecular Forces and Molecular Weights:
- Methane: Lowest molecular weight and only weak LDFs.
- Ethanol: Highest molecular weight amongst the set, and strong hydrogen bonding, which significantly elevates its boiling point.
- Chloromethane: Intermediate molecular weight and polar, having both dipole-dipole forces and LDFs.
- Propane: Intermediate molecular weight and nonpolar, only experiencing LDFs.
Step 4: Determine the Lowest Boiling Point:
Based on the analysis, methane (CH₄) will have the lowest boiling point. It has the lowest molecular weight and only experiences weak London Dispersion Forces, requiring the least amount of energy to overcome the IMFs and transition to the gaseous phase.
Advanced Considerations and Exceptions
While the above approach provides a general framework, several nuances can influence boiling point predictions:
-
Branching: As mentioned earlier, branched molecules tend to have lower boiling points than their linear isomers due to reduced surface area for interaction and weaker LDFs.
-
Isomerism: Different isomers of the same compound can have different boiling points depending on their shapes and the resulting strength of their IMFs.
-
Intramolecular Hydrogen Bonding: In some instances, intramolecular hydrogen bonding (hydrogen bonding within the same molecule) can reduce the effectiveness of intermolecular hydrogen bonding, leading to lower boiling points than expected.
-
Complex Molecules: Predicting boiling points for large, complex molecules becomes significantly more challenging, often requiring computational methods and specialized software.
Practical Applications and Further Exploration
Understanding the relationship between IMFs and boiling points is crucial in numerous fields:
-
Chemistry: Designing and synthesizing new compounds with specific boiling points for applications such as solvents, refrigerants, and pharmaceuticals.
-
Chemical Engineering: Optimizing separation processes like distillation, relying on differences in boiling points to isolate components of a mixture.
-
Materials Science: Developing materials with desirable properties by manipulating IMFs.
This article offers a solid foundation for understanding how to determine which compound has the lowest boiling point. Remember to carefully consider all factors – molecular weight, molecular shape, polarity, and the presence of hydrogen bonding – to accurately predict boiling point trends. Further exploration into advanced topics like computational chemistry and thermodynamic principles will enhance your understanding and predictive capabilities. Continuously refining your knowledge of IMFs and their impact on physical properties will improve your ability to solve increasingly complex chemical problems.
Latest Posts
Latest Posts
-
A Pendulum Consists Of A Small Object
Apr 06, 2025
-
Fertilization In Humans Normally Occurs In The
Apr 06, 2025
-
What Is The Ph Of A 0 001 M Koh Solution
Apr 06, 2025
-
What Is 0 6 As A Fraction In Simplest Form
Apr 06, 2025
-
Write Iupac Name Of The Following Compound
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Compounds Has The Lowest Boiling Point . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
