Sodium Sulfate Barium Chloride Balanced Equation
News Leon
Apr 04, 2025 · 6 min read
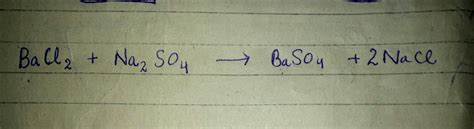
Table of Contents
Sodium Sulfate and Barium Chloride: A Deep Dive into the Balanced Equation and its Implications
The reaction between sodium sulfate (Na₂SO₄) and barium chloride (BaCl₂) is a classic example of a double displacement or metathesis reaction, frequently used in chemistry demonstrations and educational contexts to illustrate concepts like precipitation reactions and stoichiometry. Understanding this reaction, including its balanced equation, the driving force behind it, and its practical applications, is crucial for a solid grasp of fundamental chemical principles.
Understanding the Reaction: A Double Displacement
The reaction between sodium sulfate and barium chloride is a double displacement reaction, meaning that the cations (positively charged ions) and anions (negatively charged ions) of the two reactants switch partners to form two new products. In this specific case:
- Reactants: Sodium sulfate (Na₂SO₄) and barium chloride (BaCl₂)
- Products: Barium sulfate (BaSO₄) and sodium chloride (NaCl)
This reaction can be represented symbolically as:
Na₂SO₄(aq) + BaCl₂(aq) → BaSO₄(s) + 2NaCl(aq)
This is the balanced equation, crucial for understanding the stoichiometric relationships between reactants and products. Let's break down why balancing is necessary and how it's achieved.
Balancing the Equation: Conservation of Mass
The principle of conservation of mass dictates that in any chemical reaction, the total mass of the reactants must equal the total mass of the products. Balancing a chemical equation ensures this principle is upheld by adjusting the stoichiometric coefficients (the numbers in front of each chemical formula) to ensure that the number of atoms of each element is the same on both sides of the equation.
In the unbalanced equation:
Na₂SO₄(aq) + BaCl₂(aq) → BaSO₄(s) + NaCl(aq)
We can see an imbalance:
- Sodium (Na): 2 atoms on the left, 1 atom on the right.
- Sulfur (S): 1 atom on the left, 1 atom on the right.
- Oxygen (O): 4 atoms on the left, 4 atoms on the right.
- Barium (Ba): 1 atom on the left, 1 atom on the right.
- Chlorine (Cl): 2 atoms on the left, 1 atom on the right.
To balance the equation, we need to add a coefficient of 2 in front of NaCl:
Na₂SO₄(aq) + BaCl₂(aq) → BaSO₄(s) + 2NaCl(aq)
Now the number of atoms of each element is equal on both sides, fulfilling the law of conservation of mass.
The Driving Force: Precipitation
The primary driving force behind this reaction is the formation of an insoluble precipitate. When aqueous solutions of sodium sulfate and barium chloride are mixed, barium sulfate (BaSO₄) precipitates out of the solution as a white solid. This precipitation is the key factor that pushes the reaction forward to completion.
The solubility of ionic compounds depends on the interactions between the ions and the solvent (water, in this case). While sodium chloride (NaCl) is highly soluble in water, barium sulfate (BaSO₄) is virtually insoluble. The formation of this insoluble solid removes barium and sulfate ions from the solution, preventing the reverse reaction from occurring significantly. This is governed by Le Chatelier's Principle, which states that a system at equilibrium will shift to counteract any stress applied to it. The stress in this case is the removal of BaSO₄ from the solution, causing the reaction to proceed further to the right.
Solubility Rules and Predicting Precipitation Reactions
Predicting whether a precipitation reaction will occur requires knowledge of solubility rules. These rules provide general guidelines on the solubility of various ionic compounds in water. While there are exceptions, they are a useful tool for predicting the outcome of reactions involving ionic compounds. In our case:
- Sulfates (SO₄²⁻): Most sulfates are soluble, except for those of barium, strontium, calcium, lead, and silver.
- Chlorides (Cl⁻): Most chlorides are soluble, except for those of silver, lead, and mercury(I).
Based on these rules, we can predict that barium sulfate will be insoluble (precipitate), while sodium chloride will remain soluble.
Applications and Practical Significance
The reaction between sodium sulfate and barium chloride, while seemingly simple, has several practical applications:
1. Qualitative Analysis: Identifying Barium Ions
The formation of the white barium sulfate precipitate can be used as a qualitative test for the presence of barium ions (Ba²⁺) in a solution. If an unknown solution is mixed with sodium sulfate and a white precipitate forms, it indicates the presence of barium ions. This is a common test in analytical chemistry.
2. Gravimetric Analysis: Determining the Amount of Barium or Sulfate
The insolubility of barium sulfate allows for its use in gravimetric analysis. In this technique, the barium sulfate precipitate is carefully filtered, dried, and weighed. Knowing the molar mass of barium sulfate, the amount of barium or sulfate ions in the original sample can be calculated. This precise method is often used in determining the concentration of sulfate in various samples, such as water or soil.
3. Preparation of Barium Sulfate: Medical and Industrial Uses
Barium sulfate is a significant product of this reaction. It has crucial applications in medicine as a contrast agent for X-ray imaging of the gastrointestinal tract. Because it is radiopaque (it absorbs X-rays), barium sulfate suspension allows for clear visualization of the digestive system. It's also used in some industrial applications as a pigment and a filler in various products. Importantly, despite barium being toxic, the insolubility of barium sulfate renders it safe for ingestion in controlled medical contexts.
Beyond the Basics: Factors Influencing the Reaction
Several factors can influence the outcome and efficiency of the reaction between sodium sulfate and barium chloride:
1. Concentration of Reactants:
Higher concentrations of reactants generally lead to a faster and more complete reaction, resulting in a larger amount of precipitate.
2. Temperature:
While the effect of temperature on the solubility of barium sulfate is relatively small, higher temperatures can slightly increase the reaction rate.
3. Presence of Other Ions:
The presence of other ions in the solution can sometimes interfere with the reaction, affecting the solubility of barium sulfate and the completeness of the precipitation.
4. Reaction Kinetics:
The rate of precipitation depends on several factors, including the collision frequency of the reacting ions and the activation energy required for the formation of the barium sulfate crystal lattice.
Conclusion: A Foundation for Chemical Understanding
The reaction between sodium sulfate and barium chloride offers a valuable case study for understanding fundamental chemical concepts, including double displacement reactions, stoichiometry, precipitation reactions, solubility rules, and gravimetric analysis. Its seemingly simple equation belies a wealth of underlying chemical principles and practical applications, making it a cornerstone of introductory and advanced chemistry studies. The balanced equation, Na₂SO₄(aq) + BaCl₂(aq) → BaSO₄(s) + 2NaCl(aq), is not just a symbolic representation; it’s a key to unlocking a deeper understanding of the quantitative and qualitative aspects of chemical reactions and their importance in various fields. Further exploration of these concepts will strengthen your understanding of chemical reactivity and its practical implications.
Latest Posts
Latest Posts
-
Any Computer Parts That You Can Actually Touch
Apr 04, 2025
-
Which Of The Following Is Hydrophobic
Apr 04, 2025
-
Do Prokaryotes Have A Membrane Bound Organelles
Apr 04, 2025
-
Why Was A Stain Added To The Cheek Cells
Apr 04, 2025
-
What Is A Group Of Tissues That Work Together Called
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Sodium Sulfate Barium Chloride Balanced Equation . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
