Does Ionization Increase From Left To Right
News Leon
Apr 04, 2025 · 6 min read
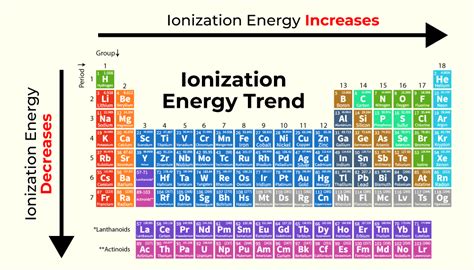
Table of Contents
Does Ionization Energy Increase from Left to Right Across a Period? A Comprehensive Exploration
Ionization energy, a fundamental concept in chemistry, dictates the energy required to remove an electron from a gaseous atom or ion. Understanding its trends across the periodic table is crucial for predicting chemical behavior and reactivity. A common question revolves around the change in ionization energy as we move from left to right across a period (a horizontal row). The short answer is yes, ionization energy generally increases from left to right across a period. However, this seemingly simple statement requires a deeper dive to fully appreciate the underlying principles and nuances. This article will explore the reasons behind this trend, examining the interplay of nuclear charge, atomic radius, and electron shielding. We will also delve into exceptions and explore the implications of this trend for various chemical properties.
The Fundamental Factors Influencing Ionization Energy
Three key factors determine the magnitude of ionization energy:
1. Nuclear Charge: The Stronger Pull
The positive charge of the nucleus exerts an attractive force on the negatively charged electrons. As we move across a period, the number of protons in the nucleus increases, leading to a stronger positive charge. This stronger nuclear pull makes it progressively more difficult to remove an electron, thus increasing the ionization energy. The increase in nuclear charge is the primary driver behind the overall trend of increasing ionization energy from left to right.
2. Atomic Radius: The Distance Matters
Atomic radius refers to the size of an atom. Across a period, the atomic radius generally decreases. This is because, while additional electrons are added to the same principal energy level (shell), the increasing nuclear charge pulls these electrons closer to the nucleus, resulting in a smaller atomic size. The closer the outermost electrons are to the nucleus, the stronger the attractive force, making it harder to remove them and hence increasing ionization energy. A smaller radius translates to a stronger electrostatic attraction between the nucleus and valence electrons.
3. Shielding Effect: The Electron Cloud's Influence
Electron shielding, also known as screening, is the effect of inner electrons reducing the attractive force of the nucleus on the outer electrons. Inner electrons partially shield the outer electrons from the full positive charge of the nucleus. While the nuclear charge increases across a period, the shielding effect from inner electrons remains relatively constant within the same shell. Therefore, the increase in nuclear charge outweighs the relatively constant shielding effect, leading to a net increase in the attraction between the nucleus and valence electrons, hence the increase in ionization energy.
The Trend in Detail: A Period-by-Period Look
Let's examine the trend in ionization energy across a period more closely, considering the nuances that may arise:
-
Period 2 (Li to Ne): The ionization energy increases steadily from lithium (Li) to neon (Ne). Lithium has a relatively low ionization energy because its single valence electron is relatively far from the nucleus and weakly held. As we progress across the period, the nuclear charge increases, the atomic radius decreases, and the increased effective nuclear charge makes it progressively more difficult to remove an electron. Neon, with its full valence shell, has the highest ionization energy in this period.
-
Period 3 (Na to Ar): The same trend applies to Period 3, with sodium (Na) having the lowest ionization energy and argon (Ar) having the highest. The effect of increased nuclear charge and decreased atomic radius dominates, leading to a steady increase in ionization energy.
-
Beyond Period 3: The trend of increasing ionization energy from left to right continues in subsequent periods, though the magnitude of the increase may vary slightly due to subtle differences in electron configurations and subtle variations in shielding effects.
Exceptions and Irregularities: Why the Trend Isn't Always Perfect
While the general trend of increasing ionization energy from left to right is observed, some irregularities exist. These deviations primarily arise due to:
-
Electron Configuration: The stability of electron configurations plays a role. For example, a half-filled or fully filled subshell provides extra stability. Removing an electron from such a stable configuration requires more energy, causing a slight increase in ionization energy compared to the expected trend.
-
Subshell Penetration: Electrons in different subshells (s, p, d, f) penetrate the electron cloud to varying degrees. Electrons in s subshells, for example, penetrate closer to the nucleus than those in p subshells. These differences in penetration affect the shielding effect and can lead to small deviations from the expected trend.
-
Inter-electronic Repulsions: The repulsion between electrons can also influence ionization energy. In some cases, repulsions between electrons in the same subshell might slightly counteract the increased nuclear charge, causing a small decrease in the ionization energy compared to the perfectly smooth increase.
Implications of the Trend: Chemical Reactivity and Properties
The trend of increasing ionization energy across a period has profound implications for the chemical properties of elements:
-
Metallic Character: Elements on the left side of a period have lower ionization energies and tend to be more metallic (easily losing electrons to form positive ions). As we move to the right, ionization energy increases, and metallic character decreases.
-
Non-metallic Character: Elements on the right side of a period have higher ionization energies and are more non-metallic (less likely to lose electrons and more likely to gain electrons to form negative ions).
-
Reactivity: Elements with low ionization energies (on the left) are generally more reactive than elements with high ionization energies (on the right). This is because they readily lose electrons to form stable ions and participate in chemical reactions.
-
Bonding: The ionization energy significantly influences the type of chemical bonds formed by an element. Elements with low ionization energies tend to form ionic bonds (transfer of electrons), while those with high ionization energies often form covalent bonds (sharing of electrons).
Conclusion: A Fundamental Trend with Subtleties
The general trend of increasing ionization energy from left to right across a period is a fundamental concept in chemistry. This trend is primarily driven by the increasing nuclear charge and decreasing atomic radius, with electron shielding playing a supporting role. While the trend is generally observed, subtle irregularities can arise due to electron configurations, subshell penetration, and electron-electron repulsions. Understanding this trend is crucial for comprehending the chemical behavior and reactivity of elements and their compounds, providing a cornerstone for further explorations in chemical bonding, periodic properties, and beyond. The interplay between these forces offers a fascinating insight into the intricate workings of atoms and their interactions, highlighting the complexity and beauty of the periodic table. Continued research in atomic structure and quantum mechanics constantly refines our understanding of these fundamental principles, further solidifying the importance of ionization energy in the realm of chemistry.
Latest Posts
Latest Posts
-
Any Computer Parts That You Can Actually Touch
Apr 04, 2025
-
Which Of The Following Is Hydrophobic
Apr 04, 2025
-
Do Prokaryotes Have A Membrane Bound Organelles
Apr 04, 2025
-
Why Was A Stain Added To The Cheek Cells
Apr 04, 2025
-
What Is A Group Of Tissues That Work Together Called
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Does Ionization Increase From Left To Right . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
