The Function Of Dna Ligase In Recombinant Technology Is To
News Leon
Apr 05, 2025 · 6 min read
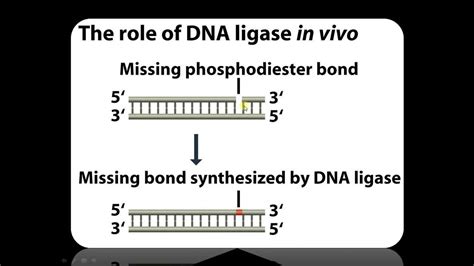
Table of Contents
The Function of DNA Ligase in Recombinant Technology Is To… Join DNA Fragments!
DNA ligase plays a pivotal role in the field of recombinant DNA technology, acting as the molecular "glue" that joins DNA fragments together. Understanding its function is crucial for anyone working with genetic engineering, gene therapy, or other applications involving the manipulation of DNA. This article delves deep into the role of DNA ligase, exploring its mechanism, various types, applications in recombinant technology, and its significance in advancing biotechnology.
Understanding DNA Ligase: The Molecular Glue
DNA ligase is an enzyme that catalyzes the formation of a phosphodiester bond between the 3'-hydroxyl group of one DNA fragment and the 5'-phosphate group of another. This process is essential for repairing breaks in DNA strands, and it's this very ability that makes it indispensable in recombinant DNA technology. Essentially, it seals the gaps between two DNA molecules, creating a continuous, stable strand. Think of it as the molecular equivalent of a sophisticated "stapler" for DNA.
The Mechanism of Action: A Closer Look
The mechanism of DNA ligase action involves several key steps. First, the enzyme recognizes and binds to the nick in the DNA strand – the break in the phosphodiester backbone. This recognition often involves specific sequences flanking the break. Subsequently, the enzyme activates the 5'-phosphate group, often using ATP (adenosine triphosphate) or NAD+ (nicotinamide adenine dinucleotide) as a cofactor, converting it into a high-energy intermediate. This activated phosphate then reacts with the 3'-hydroxyl group of the adjacent DNA fragment, forming the crucial phosphodiester bond and sealing the nick. The enzyme then dissociates from the newly ligated DNA.
Different types of DNA ligases utilize different cofactors and have varying mechanisms for activating the 5'-phosphate, but the overall goal remains the same: to create a stable phosphodiester bond between two DNA fragments.
Types of DNA Ligases and Their Applications
Various types of DNA ligases exist, each with its unique properties and applications in recombinant DNA technology. The most commonly used are:
1. T4 DNA Ligase: The Workhorse of Recombinant DNA Technology
T4 DNA ligase, isolated from the T4 bacteriophage, is arguably the most widely used DNA ligase in molecular biology. It's known for its efficiency in joining blunt-ended DNA fragments, although it works more efficiently with cohesive (sticky) ends generated by restriction enzyme digestion. Its robustness and relatively high activity make it a staple in many molecular biology laboratories. Its preference for ATP as a cofactor also contributes to its widespread use.
2. E. coli DNA Ligase: Specific Needs, Specific Enzyme
E. coli DNA ligase, derived from Escherichia coli, shows a preference for NAD+ as a cofactor, unlike T4 DNA ligase. This difference can influence its application in specific experimental setups. Furthermore, its activity is generally higher with nicked DNA than with blunt-ended DNA. This characteristic can be advantageous in some cloning procedures but less so in others.
3. Other DNA Ligases: Specialized Roles
Other DNA ligases exist, derived from various organisms, each exhibiting unique properties that may be suited to specific applications. These specialized ligases may exhibit higher activity with specific DNA sequences or under specific reaction conditions, making them valuable tools for tailored genetic engineering projects.
DNA Ligase: A Crucial Tool in Recombinant DNA Technology
DNA ligase's importance in recombinant DNA technology cannot be overstated. Its role extends across numerous crucial techniques, including:
1. Cloning: Joining DNA Fragments into Vectors
This is arguably the most important application. Recombinant DNA technology often involves inserting a gene of interest (e.g., a human gene encoding a therapeutic protein) into a vector, a DNA molecule capable of self-replication within a host cell (e.g., a plasmid or a viral vector). DNA ligase is the essential enzyme that seals the gene of interest into the vector, creating a recombinant DNA molecule. This step is fundamental for creating genetically modified organisms (GMOs), producing recombinant proteins, and numerous other applications.
2. Site-Directed Mutagenesis: Precise Modifications of DNA Sequences
This technique allows for specific changes to a DNA sequence, including introducing mutations, deleting sections, or inserting new sequences. DNA ligase is used to join the modified DNA fragments back together, creating a DNA molecule with the desired changes. This is crucial for studying gene function, engineering proteins with altered properties, and developing new therapies.
3. DNA Sequencing: Preparing Samples for Analysis
While not directly involved in the sequencing reaction itself, DNA ligase can play a supporting role. In some sequencing methods, DNA ligase is used to join adapters or linkers to DNA fragments, preparing them for efficient sequencing.
4. Gene Therapy: Delivering Therapeutic Genes
Gene therapy aims to treat genetic disorders by introducing functional genes into a patient's cells. DNA ligase is often involved in constructing the vectors used to deliver these therapeutic genes. The precision of DNA ligase in joining DNA fragments is essential for ensuring the correct insertion of the therapeutic gene into the vector.
5. Gene Synthesis: Constructing Artificial Genes
Scientists are increasingly able to synthesize genes from scratch. DNA ligase plays a critical role in this process, joining short, synthetic DNA oligonucleotides to build longer genes. This allows for the creation of novel genes with desired properties.
6. Genome Editing: Precise Alterations of Genomes
CRISPR-Cas9 and other genome editing technologies utilize DNA ligase indirectly. While the Cas9 enzyme creates a double-stranded break, DNA ligase is often involved in the subsequent repair process, either via non-homologous end joining (NHEJ) or homology-directed repair (HDR), depending on the context. The efficiency of these repair pathways can impact the success of genome editing experiments.
Optimizing DNA Ligase Activity for Enhanced Efficiency
Several factors can influence the efficiency of DNA ligase, including:
-
DNA concentration: An optimal concentration of DNA is essential for efficient ligation. Too little DNA results in a lower probability of successful ligation, while too much can lead to non-specific ligation events.
-
ATP or NAD+ concentration: The availability of the appropriate cofactor (ATP for T4 DNA ligase, NAD+ for E. coli DNA ligase) is critical. Insufficient cofactor will limit the enzyme's activity.
-
Temperature: The optimal temperature for DNA ligase activity varies depending on the specific enzyme. Incubating the reaction at the optimal temperature is crucial for efficient ligation.
-
Buffer conditions: The ionic strength and pH of the reaction buffer influence DNA ligase activity. Using the appropriate buffer is essential for optimal performance.
-
Presence of inhibitors: Certain substances can inhibit DNA ligase activity. Care must be taken to avoid these inhibitors during the ligation reaction.
-
DNA end structure: As mentioned earlier, T4 DNA ligase works more efficiently with cohesive (sticky) ends. Blunt-end ligation generally requires higher enzyme concentrations and longer incubation times.
Conclusion: An Indispensable Tool in Biotechnology
DNA ligase, with its remarkable ability to join DNA fragments, stands as a cornerstone of recombinant DNA technology. Its diverse applications in cloning, site-directed mutagenesis, gene therapy, gene synthesis, and genome editing highlight its importance in advancing biotechnology. Continued research into DNA ligases and their mechanisms of action promises even more sophisticated applications in the future, opening new doors in genetic engineering, medicine, and other fields. A thorough understanding of its function and optimization strategies is crucial for researchers seeking to harness its power in various biotechnological endeavors. The precise and efficient joining of DNA fragments provided by this vital enzyme continues to shape our understanding and manipulation of the genetic code.
Latest Posts
Latest Posts
-
How Many Chromosomes Will Be In Each Daughter Cell
Apr 06, 2025
-
One Feature Of Pure Monopoly Is That The Firm Is
Apr 06, 2025
-
What Is The Equivalent Capacitance Of The Combination Shown
Apr 06, 2025
-
What Is The Area Of The Triangle Shown
Apr 06, 2025
-
Why Are Fresh Vegetables Sprinkled With Water At Markets
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about The Function Of Dna Ligase In Recombinant Technology Is To . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
