How Many Electrons Are In Mg 2
News Leon
Apr 02, 2025 · 5 min read
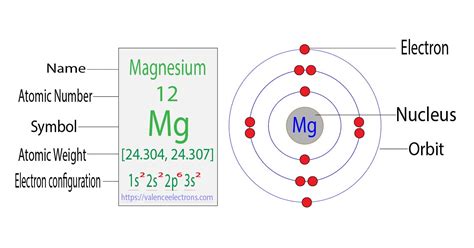
Table of Contents
How Many Electrons Are in Mg²⁺? Understanding Ionic Charges and Electron Configuration
Magnesium (Mg), a vital element for human health and numerous industrial applications, often exists as a cation with a 2+ charge (Mg²⁺). Understanding the number of electrons in this ion requires a grasp of fundamental chemistry concepts like electron shells, valence electrons, and ionic bonding. This article delves deep into the electron configuration of magnesium and its ion, explaining the process of ionization and the significance of this charged species.
Understanding Magnesium's Atomic Structure
Before we tackle the Mg²⁺ ion, let's establish the basic structure of a neutral magnesium atom. Magnesium, with an atomic number of 12, possesses 12 protons in its nucleus. In a neutral atom, the number of protons equals the number of electrons, maintaining electrical neutrality. Therefore, a neutral magnesium atom has 12 electrons.
Electron Shell Arrangement
These 12 electrons are arranged in specific energy levels or shells around the nucleus. Following the Aufbau principle, which dictates that electrons fill lower energy levels first, the electron configuration of magnesium is:
- 1s² 2s² 2p⁶ 3s²
Let's break this down:
- 1s²: Two electrons occupy the first shell (n=1), in the 's' orbital.
- 2s² 2p⁶: Eight electrons occupy the second shell (n=2). Two electrons fill the 's' orbital, and six fill the three 'p' orbitals.
- 3s²: The remaining two electrons reside in the third shell (n=3), in the 's' orbital.
These outermost electrons in the 3s orbital are called valence electrons. They are the electrons most involved in chemical bonding. Magnesium's two valence electrons determine its chemical reactivity.
The Formation of Mg²⁺: Ionization and Electron Loss
Magnesium readily loses its two valence electrons to achieve a stable electron configuration, mimicking that of a noble gas (Neon). This process is called ionization. The loss of these electrons results in the formation of a positively charged ion, Mg²⁺.
The Role of Valence Electrons in Ionization
The valence electrons are loosely held by the nucleus compared to the inner electrons. This makes them easier to remove. The energy required to remove an electron is called the ionization energy. Magnesium has relatively low ionization energies for its first two electrons, making it energetically favorable for it to lose them.
The Stable Octet: Achieving Noble Gas Configuration
By losing its two valence electrons, magnesium achieves a stable electron configuration, identical to that of Neon (1s² 2s² 2p⁶). This configuration, with a filled outer shell of eight electrons (an octet), is exceptionally stable. Atoms tend to gain, lose, or share electrons to achieve this stable state.
Determining the Number of Electrons in Mg²⁺
Since magnesium loses two electrons to become Mg²⁺, the number of electrons in the ion is simply the original number of electrons minus the number lost.
- Original number of electrons (Mg): 12
- Electrons lost: 2
- Number of electrons in Mg²⁺: 12 - 2 = 10
Therefore, the Mg²⁺ ion contains 10 electrons.
Electron Configuration of Mg²⁺
The electron configuration of Mg²⁺ reflects the loss of the two 3s electrons:
- 1s² 2s² 2p⁶
This configuration is isoelectronic with Neon, meaning it has the same number of electrons as Neon.
Significance of Mg²⁺ in Chemical Reactions and Biological Systems
The Mg²⁺ ion plays a crucial role in a wide range of chemical reactions and biological processes. Its 2+ charge allows it to participate in ionic bonding with negatively charged ions or molecules.
Ionic Bonding and Crystal Structures
Mg²⁺ readily forms ionic bonds with anions such as chloride (Cl⁻) to form ionic compounds like magnesium chloride (MgCl₂). In these compounds, the electrostatic attraction between the positively charged Mg²⁺ and negatively charged Cl⁻ ions holds the crystal lattice together. The strong electrostatic forces contribute to the high melting and boiling points of these ionic compounds.
Biological Roles of Magnesium Ions
Magnesium ions are essential for numerous biological functions:
- Enzyme Activation: Mg²⁺ acts as a cofactor for many enzymes, meaning it's necessary for their proper functioning. It participates in various metabolic processes, including DNA replication and protein synthesis.
- Muscle Contraction: Mg²⁺ plays a role in muscle contraction and relaxation.
- Nerve Impulse Transmission: It contributes to nerve impulse transmission.
- Bone Formation: Magnesium is a crucial component of bone structure.
The abundance and versatility of Mg²⁺ in biological systems highlight its importance for life.
Beyond Mg²⁺: Exploring Other Ions and Their Electron Configurations
The principle of determining the number of electrons in ions applies to other elements as well. For example:
- Na⁺ (Sodium ion): Sodium (Na) has 11 electrons. It loses one electron to form Na⁺, resulting in 10 electrons.
- Ca²⁺ (Calcium ion): Calcium (Ca) has 20 electrons. It loses two electrons to form Ca²⁺, resulting in 18 electrons.
- Al³⁺ (Aluminum ion): Aluminum (Al) has 13 electrons. It loses three electrons to form Al³⁺, resulting in 10 electrons.
Understanding the electron configurations of these ions is critical for comprehending their chemical behavior and biological roles.
Conclusion: The Importance of Understanding Ionic Charges and Electron Configuration
Determining the number of electrons in Mg²⁺, and other ions, is fundamental to understanding chemical bonding, reactivity, and the properties of matter. The loss of valence electrons to achieve a stable octet is a driving force behind the formation of ions, influencing their interactions in chemical reactions and their roles in biological systems. The principles discussed here apply broadly to a wide range of elements and ions, showcasing the interconnectedness of atomic structure and chemical behavior. This knowledge forms a cornerstone of chemistry and is crucial for understanding the world around us.
Latest Posts
Latest Posts
-
Which Of The Following Compounds Are Aromatic
Apr 03, 2025
-
An Organic Molecule Will Always Contain
Apr 03, 2025
-
Which Of The Following Is Not Homogeneous Mixture
Apr 03, 2025
-
True Or False All Whole Numbers Are Rational Numbers
Apr 03, 2025
-
What Is The Product Of Any Integer And 1
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Electrons Are In Mg 2 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
