Which Of The Following Is Not Homogeneous Mixture
News Leon
Apr 03, 2025 · 5 min read
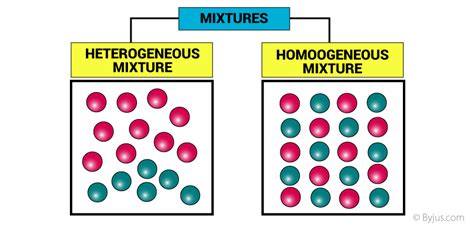
Table of Contents
Which of the Following is NOT a Homogeneous Mixture? Understanding Mixtures and Their Classifications
Understanding the difference between homogeneous and heterogeneous mixtures is fundamental to chemistry and many other scientific disciplines. While the definition seems straightforward, the nuances can be tricky. This article delves deep into the classification of mixtures, focusing specifically on identifying which substances are not homogeneous mixtures. We'll explore various examples, explain the underlying principles, and provide practical tips for identifying mixture types.
What is a Homogeneous Mixture?
A homogeneous mixture is a type of mixture where the composition is uniform throughout the mixture. This means that at a macroscopic level (visible to the naked eye), the different components of the mixture are indistinguishable from one another. No matter where you take a sample from the mixture, its composition will be identical. Think of it like a perfectly blended smoothie – you won't see individual pieces of fruit.
Key characteristics of homogeneous mixtures:
- Uniform composition: The components are evenly distributed throughout.
- Single phase: The mixture exists in a single phase (solid, liquid, or gas). You won't see distinct layers or separate components.
- Particle size: The particles of the components are incredibly small, typically at the atomic or molecular level. This ensures a uniform distribution.
What is a Heterogeneous Mixture?
In contrast to a homogeneous mixture, a heterogeneous mixture has a non-uniform composition. This means the different components are visible and easily distinguishable from one another. If you were to take samples from different parts of the mixture, their compositions would vary. Think of a salad – you can clearly see the individual pieces of lettuce, tomato, and cucumber.
Key characteristics of heterogeneous mixtures:
- Non-uniform composition: The components are not evenly distributed.
- Multiple phases: The mixture may exist in multiple phases (solid, liquid, or gas). You will see distinct layers or separate components.
- Particle size: The particles of the components are larger and easily visible.
Examples of Homogeneous Mixtures
Many everyday substances are homogeneous mixtures. These include:
- Air: A mixture of gases like nitrogen, oxygen, carbon dioxide, and others. Despite the presence of multiple gases, air appears uniform to the naked eye.
- Saltwater: When salt (NaCl) is dissolved completely in water (H₂O), it forms a homogeneous mixture. The salt ions are evenly distributed throughout the water.
- Sugar water: Similar to saltwater, dissolving sugar in water creates a homogeneous mixture.
- Steel: An alloy of iron and carbon, steel is a solid homogeneous mixture.
- Brass: Another alloy, brass is a homogeneous mixture of copper and zinc.
- Vinegar: A solution of acetic acid in water.
- Most commercially available soft drinks: Although composed of multiple substances, the final mixture is visually uniform.
Examples of Heterogeneous Mixtures
Heterogeneous mixtures are often easily identified by their visible components. Examples include:
- Sand and water: The sand particles are clearly visible and separate from the water.
- Oil and water: These two liquids do not mix, resulting in distinct layers.
- Salad: As mentioned before, a salad contains clearly distinguishable ingredients.
- Pizza: A pizza contains various toppings that are not evenly distributed.
- Concrete: A mixture of cement, sand, gravel, and water. The different components are visible.
- Granite: A rock composed of different minerals, each easily identifiable.
- Blood: Although appearing homogeneous at first glance, blood is actually a heterogeneous mixture with different cells suspended in plasma.
Identifying Non-Homogeneous Mixtures: A Detailed Approach
To determine if a mixture is not homogeneous (i.e., it's heterogeneous), you need to carefully examine its properties. Here's a step-by-step approach:
- Visual Inspection: The simplest method. Can you see distinct components or phases? If yes, it's likely heterogeneous.
- Microscopic Examination: For mixtures where visual inspection isn't conclusive, a microscope can reveal the presence of different components at a smaller scale.
- Filtration: If you can physically separate components using a filter, it's a heterogeneous mixture. For example, you can filter sand out of sand and water.
- Centrifugation: This technique separates components based on density. If centrifugation yields separate layers, the mixture is heterogeneous. This is often used to separate blood components.
- Sedimentation: Over time, some heterogeneous mixtures will settle, with denser components sinking to the bottom.
- Tyndall Effect: This effect occurs when a light beam is shone through a mixture. If the beam is visible due to scattering by particles, the mixture is likely heterogeneous (colloid). This is often used to distinguish between solutions (homogeneous) and colloids (heterogeneous).
Commonly Confused Mixtures: Solutions, Suspensions, and Colloids
The terms solution, suspension, and colloid are often used to describe mixtures, and it's crucial to understand their differences, especially regarding homogeneity.
-
Solutions: These are always homogeneous mixtures. The solute particles are completely dissolved and evenly distributed at a molecular level. Examples include saltwater and sugar water.
-
Suspensions: These are heterogeneous mixtures where the particles are large enough to settle out over time. Examples include sand and water or muddy water.
-
Colloids: These are in between solutions and suspensions. They appear homogeneous at first glance, but the dispersed particles are larger than in solutions, and they scatter light (Tyndall effect). Examples include milk, fog, and smoke. While they might appear homogeneous, they are technically heterogeneous because of the particle size and scattering of light.
Advanced Considerations: Scale and Observation
The determination of homogeneity can depend on the scale of observation. A mixture that appears homogeneous at the macroscopic level might be heterogeneous at a microscopic level. For example, a seemingly uniform sample of soil might contain diverse components when examined under a microscope. Therefore, the context of the observation is critical.
Conclusion: Homogeneity Requires Uniformity at All Levels
Determining whether a mixture is homogeneous or heterogeneous requires a comprehensive analysis considering both macroscopic and microscopic properties. While visual inspection is a good starting point, more advanced techniques may be necessary for mixtures where the distinction is less obvious. The critical aspect is uniformity – a true homogeneous mixture displays uniform composition at every level of observation. Any deviation from this uniformity signifies a heterogeneous mixture. By understanding these principles and employing the techniques described above, you can confidently classify any mixture and understand its unique properties.
Latest Posts
Latest Posts
-
Collection Of Similar Cells That Perform A Particular Function
Apr 03, 2025
-
Which Atomic Particle Determines The Chemical Behavior Of An Atom
Apr 03, 2025
-
Which Of The Following Occurs First Causing Expiration
Apr 03, 2025
-
A Homogeneous Mixture Is Also Called A
Apr 03, 2025
-
Least Common Multiple Of 3 6 9
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is Not Homogeneous Mixture . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
