Which Of The Following Compounds Are Aromatic
News Leon
Apr 03, 2025 · 6 min read
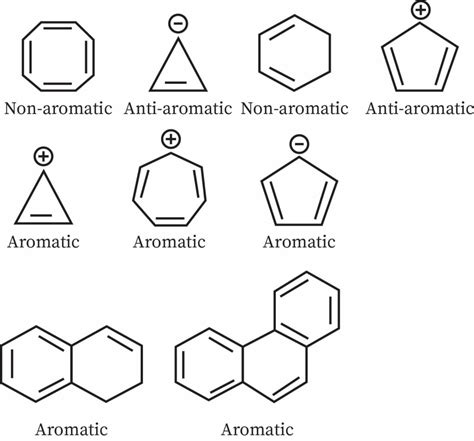
Table of Contents
Which of the Following Compounds are Aromatic? A Deep Dive into Aromaticity
Aromatic compounds are a fascinating class of organic molecules with unique properties stemming from their cyclic, planar structure and a specific number of pi electrons. Understanding aromaticity is crucial for comprehending the reactivity and stability of countless organic compounds. This article will delve into the criteria for aromaticity, exploring several examples and non-examples to solidify your understanding. We'll examine various compounds and definitively determine whether they meet the rigorous requirements for aromatic character.
The Huckel's Rule: The Cornerstone of Aromaticity
The foundation of aromatic character lies in Hückel's rule, which states that a planar, cyclic, conjugated molecule is aromatic if it contains (4n + 2) π electrons, where 'n' is a non-negative integer (n = 0, 1, 2, 3...). This magic number of electrons allows for the delocalization of the π electrons across the entire ring system, resulting in enhanced stability. Let's break down the three essential criteria:
1. Cyclic: The Ring Requirement
The molecule must possess a closed ring structure. Open-chain conjugated systems, no matter how many π electrons they possess, are not considered aromatic.
2. Planar: The Flat Structure
Planarity is crucial. The p-orbitals of all the atoms in the ring must be able to overlap effectively. Any deviation from planarity disrupts the continuous delocalization of electrons and negates aromaticity.
3. Conjugated: The Continuous Pi System
The molecule must exhibit continuous conjugation. This means that there must be an uninterrupted series of overlapping p-orbitals around the ring. Single bonds interspersed within the ring can participate in conjugation as long as they are adjacent to double bonds (or other p-orbital containing atoms like nitrogen or oxygen).
4. (4n + 2) π Electrons: The Electron Count
The most crucial criterion. The molecule must possess a total number of π electrons that fits Hückel's (4n + 2) rule. Let's look at some examples:
- n = 0: (4(0) + 2) = 2 π electrons (e.g., benzene)
- n = 1: (4(1) + 2) = 6 π electrons (e.g., benzene)
- n = 2: (4(2) + 2) = 10 π electrons (e.g., naphthalene)
- n = 3: (4(3) + 2) = 14 π electrons (e.g., anthracene)
Analyzing Compounds for Aromaticity: Examples and Non-Examples
Now, let's dissect several examples to determine if they are aromatic based on Hückel's rule and the three essential criteria.
Example 1: Benzene (C₆H₆)
Benzene is the quintessential aromatic compound. It is cyclic, planar, and has a continuous conjugated system. Crucially, it contains six π electrons (n = 1, 4n + 2 = 6), satisfying Hückel's rule. Therefore, benzene is aromatic.
Example 2: Cyclooctatetraene (C₈H₈)
Cyclooctatetraene is a cyclic and conjugated molecule. However, it is not planar. The molecule adopts a tub-shaped conformation to minimize angle strain. This non-planarity prevents effective p-orbital overlap, meaning the π electrons are not delocalized. Furthermore, it has eight π electrons, which does not fit Hückel's rule (4n + 2). Thus, cyclooctatetraene is not aromatic. It is considered an anti-aromatic compound, possessing higher energy than expected due to electron-electron repulsion in the partially delocalized π system.
Example 3: Pyridine (C₅H₅N)
Pyridine is a six-membered ring containing five carbon atoms and one nitrogen atom. The nitrogen atom contributes one electron to the π system. Pyridine is cyclic, planar, and conjugated. It possesses six π electrons (4n + 2 where n = 1), satisfying Hückel's rule. Therefore, pyridine is aromatic.
Example 4: Furan (C₄H₄O)
Furan is a five-membered ring containing four carbon atoms and one oxygen atom. The oxygen atom contributes two electrons to the π system (one lone pair is involved in resonance). Furan is cyclic, planar, and conjugated. It has six π electrons (4n + 2 where n=1), fulfilling Hückel's rule. Consequently, furan is aromatic.
Example 5: Thiophene (C₄H₄S)
Thiophene is analogous to furan, with a sulfur atom replacing the oxygen. The sulfur atom contributes two electrons to the π system, resulting in six π electrons. It is cyclic, planar, and conjugated. Similar to furan, it meets Hückel's rule and is thus aromatic.
Example 6: Cyclobutadiene (C₄H₄)
Cyclobutadiene is a four-membered ring with alternating single and double bonds. It is cyclic and has a conjugated system if planar. However, it's highly strained and not truly planar in its ground state. More importantly, it possesses four π electrons, which does not satisfy Hückel's rule. Cyclobutadiene is anti-aromatic. Anti-aromatic compounds are less stable than expected due to the significant electron-electron repulsion between the partially delocalized π electrons.
Example 7: Azulene (C₁₀H₈)
Azulene is a fascinating example of a non-benzenoid aromatic compound. It's a fused five- and seven-membered ring system. It's planar and has a fully conjugated system. Importantly, it contains 10 π electrons (4n + 2 where n = 2), fulfilling Hückel's rule. Azulene is aromatic. Its unique structure results in a dipole moment, unlike many other aromatic systems.
Example 8: Annulenes
Annulenes are monocyclic conjugated hydrocarbons with the general formula (CH)ₙ. Their aromaticity depends solely on their size and whether they follow Hückel's rule. For instance, [10]annulene, with 10 pi electrons (n=2), could be aromatic if it were planar, but steric hindrance prevents planarity. Similarly, [14]annulene and [18] annulene can exhibit aromaticity, while those with 4n pi electrons are anti-aromatic.
Beyond Hückel's Rule: Exceptions and Nuances
While Hückel's rule serves as a powerful guideline, it's not an infallible predictor of aromaticity. Some exceptions exist:
- Non-benzenoid aromatic compounds: Azulene, as mentioned above, is a key example. Many other compounds with non-six-membered rings exhibit aromatic characteristics.
- Influence of heteroatoms: The presence of heteroatoms (atoms other than carbon) significantly impacts aromaticity, as seen in pyridine, furan, and thiophene. Their electron-donating or withdrawing capabilities affect electron distribution within the ring.
- Strain and steric factors: Ring strain can distort the planarity of the molecule, affecting the degree of conjugation and consequently, aromaticity.
Conclusion: Aromatic Compounds in Context
Understanding aromaticity is fundamental to organic chemistry. The concepts of Hückel's rule, planarity, conjugation, and the (4n + 2) electron count are crucial for predicting the stability and reactivity of organic molecules. While Hückel's rule provides a useful framework, one must always consider the structural nuances and potential exceptions. By carefully analyzing the cyclic, planar, conjugated nature of a compound and its π electron count, you can accurately determine its aromatic character and subsequently, predict its chemical behavior. This knowledge is essential in various fields, from drug discovery and materials science to organic synthesis and polymer chemistry. The world of aromatic compounds is vast and intricate, inviting further exploration and deeper understanding.
Latest Posts
Latest Posts
-
Are All Right Isosceles Triangles Similar
Apr 03, 2025
-
Do Converging Lenses Produce Virtual Images
Apr 03, 2025
-
Collection Of Similar Cells That Perform A Particular Function
Apr 03, 2025
-
Which Atomic Particle Determines The Chemical Behavior Of An Atom
Apr 03, 2025
-
Which Of The Following Occurs First Causing Expiration
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Compounds Are Aromatic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
