Arrange The Following Radicals In Order Of Increasing Stability.
News Leon
Apr 03, 2025 · 6 min read
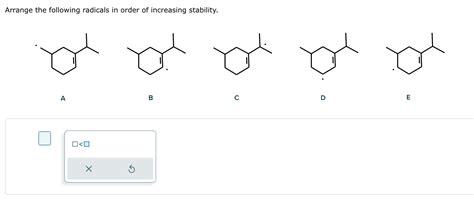
Table of Contents
Arranging Radicals in Order of Increasing Stability: A Comprehensive Guide
Understanding the stability of radicals is crucial in organic chemistry. Radicals, by definition, possess an unpaired electron, making them highly reactive. However, not all radicals are created equal. Their stability varies significantly based on several factors, and predicting this stability is a key skill for any chemist. This article delves into the factors influencing radical stability and provides a detailed guide on arranging radicals in order of increasing stability. We'll explore various types of radicals and illustrate their relative stability through numerous examples.
Factors Affecting Radical Stability
Several factors contribute to a radical's stability. The most important ones are:
-
Resonance Stabilization: Radicals with resonance structures are significantly more stable. The unpaired electron can delocalize across multiple atoms, reducing the overall electron density at any single atom. This delocalization lowers the energy of the radical, increasing its stability. Aromatic radicals, for instance, exhibit exceptional stability due to extensive resonance.
-
Hyperconjugation: This involves the interaction between the unpaired electron and the sigma bonding electrons of adjacent C-H bonds. The more alkyl groups attached to the carbon atom bearing the unpaired electron, the greater the hyperconjugative stabilization. This is why tertiary radicals are more stable than secondary, which are more stable than primary radicals.
-
Inductive Effect: Electron-donating groups (like alkyl groups) stabilize radicals through inductive effects. These groups push electron density towards the radical center, partially neutralizing the unpaired electron's charge and thus stabilizing the radical. Electron-withdrawing groups have the opposite effect, destabilizing the radicals.
-
Steric Hindrance: Bulky groups around the radical center can cause steric hindrance, destabilizing the radical. This effect is less significant than resonance or hyperconjugation but can still influence the overall stability in certain cases.
Types of Radicals and Their Relative Stability
Let's explore various types of radicals and their relative stability:
1. Methyl Radical (·CH₃): This is a primary radical. It has minimal stabilization, relying only on the inductive effect of the three hydrogen atoms.
2. Primary Radicals (RCH₂·): These radicals have one alkyl group attached to the carbon atom bearing the unpaired electron. They are more stable than methyl radicals due to the increased inductive effect of the alkyl group. However, their stability is still relatively low compared to secondary or tertiary radicals.
3. Secondary Radicals (R₂CH·): These radicals have two alkyl groups attached to the carbon atom with the unpaired electron. The increased hyperconjugation and inductive effects from the two alkyl groups make them significantly more stable than primary radicals.
4. Tertiary Radicals (R₃C·): These radicals have three alkyl groups attached to the carbon atom carrying the unpaired electron. They are the most stable alkyl radicals due to the maximum hyperconjugation and inductive stabilization.
5. Allylic Radicals: These radicals have the unpaired electron on a carbon atom adjacent to a carbon-carbon double bond. They exhibit significant resonance stabilization, making them considerably more stable than tertiary alkyl radicals.
6. Benzylic Radicals: Similar to allylic radicals, benzylic radicals have the unpaired electron on a carbon atom attached to a benzene ring. They possess extensive resonance stabilization due to the delocalization of the unpaired electron across the aromatic ring, making them exceptionally stable.
7. Vinylic Radicals: These radicals have the unpaired electron on a carbon atom of a carbon-carbon double bond (sp²-hybridized carbon). They are relatively less stable than allylic radicals due to the lack of hyperconjugation. The unpaired electron interacts with the p-orbital of the double bond but this stabilization is less than resonance stabilization in allylic radicals.
8. Aryl Radicals: These radicals have the unpaired electron on a carbon atom of an aromatic ring. Similar to benzylic radicals, they exhibit resonance stabilization but the extent of stabilization can vary depending on the substituents on the ring.
Arranging Radicals in Order of Increasing Stability: A Practical Approach
To arrange radicals in order of increasing stability, consider the factors discussed above. A general order would be:
Least Stable: Methyl Radical < Primary Radical < Secondary Radical < Tertiary Radical < Vinylic Radical < Allylic Radical < Benzylic Radical < Aryl Radical Most Stable
This is a general guideline. Specific examples may deviate slightly depending on the nature and number of substituents. For instance, a highly substituted tertiary radical might be less stable than a relatively unsubstituted allylic radical due to steric hindrance.
Example Scenarios and Detailed Analysis
Let's analyze a few examples to reinforce our understanding:
Scenario 1: Arrange the following radicals in order of increasing stability: CH₃·, (CH₃)₂CH·, CH₂=CH·, (CH₃)₃C·, C₆H₅CH₂·.
Solution:
CH₃·(Methyl Radical): Least stable due to lack of stabilization.(CH₃)₂CH·(Secondary Radical): More stable than methyl due to hyperconjugation.(CH₃)₃C·(Tertiary Radical): More stable than secondary due to increased hyperconjugation.CH₂=CH·(Vinylic Radical): Less stable than tertiary but more stable than secondary and primary due to some resonance stabilization with the double bond, although less effective than in allylic radicals.C₆H₅CH₂·(Benzylic Radical): Most stable due to extensive resonance stabilization with the benzene ring.
Therefore, the order is: CH₃· < (CH₃)₂CH· < CH₂=CH· < (CH₃)₃C· < C₆H₅CH₂·.
Scenario 2: Compare the stability of a simple tertiary alkyl radical and an allylic radical.
Solution: Generally, an allylic radical will be more stable than a tertiary alkyl radical because resonance stabilization surpasses the hyperconjugative stabilization of the tertiary radical. However, the specific substituents on both radicals can influence this order. Highly branched tertiary radicals might experience significant steric hindrance, reducing their stability and potentially placing them below less hindered allylic radicals.
Scenario 3: Consider the effect of substituents on radical stability.
Solution: Electron-donating groups (e.g., alkyl groups) stabilize radicals, while electron-withdrawing groups (e.g., halogens) destabilize them. For instance, a radical with multiple alkyl substituents will be more stable than a radical with halogen substituents.
Conclusion: Mastering Radical Stability
Understanding the factors influencing radical stability is critical in predicting reaction pathways and outcomes in organic chemistry. By carefully considering resonance, hyperconjugation, inductive effects, and steric factors, one can confidently arrange radicals in order of increasing stability. The examples and scenarios discussed above provide a strong foundation for mastering this crucial concept, enabling more accurate predictions in various organic reactions involving radical intermediates. Remember that while general trends exist, the specific arrangement can be affected by nuanced interactions of different substituents, emphasizing the importance of a case-by-case analysis for accurate prediction of radical stability. This detailed understanding enhances your proficiency in organic chemistry and allows for more effective problem-solving in this exciting and complex field.
Latest Posts
Latest Posts
-
A Fluids Resistance To Flow Is Called
Apr 04, 2025
-
Match The Location With The Appropriate Epithelial Tissue
Apr 04, 2025
-
Sodium Sulfate Barium Chloride Balanced Equation
Apr 04, 2025
-
Which Of The Following Compounds Has The Lowest Boiling Point
Apr 04, 2025
-
Does Ionization Increase From Left To Right
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Arrange The Following Radicals In Order Of Increasing Stability. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
