Arrange The Compounds In Order Of Increasing Acidity.
News Leon
Apr 05, 2025 · 6 min read
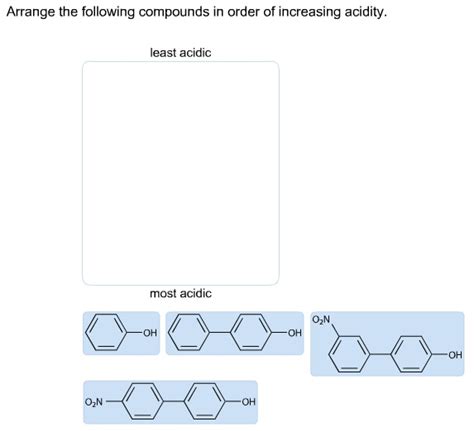
Table of Contents
Arranging Compounds in Order of Increasing Acidity: A Comprehensive Guide
Understanding acidity is fundamental in chemistry, impacting various fields from biochemistry to environmental science. The ability to arrange compounds in order of increasing acidity requires a deep understanding of several key factors that influence a molecule's tendency to donate a proton (H⁺). This comprehensive guide will delve into these factors, providing a structured approach to tackling this common chemistry challenge.
Factors Affecting Acidity
Several factors contribute to a molecule's acidity. These include:
1. Electronegativity: The Power of Electron Withdrawal
Electronegativity measures an atom's ability to attract electrons towards itself. Highly electronegative atoms, such as oxygen, fluorine, chlorine, and bromine, strongly pull electron density away from the O-H bond in an acid. This weakens the O-H bond, making it easier to donate a proton. The more electronegative atoms present and the closer they are to the acidic proton, the stronger the acid.
Example: Compare CH₃OH (methanol) and CF₃OH (trifluoromethanol). The three fluorine atoms in trifluoromethanol are highly electronegative and exert a strong electron-withdrawing inductive effect. This significantly weakens the O-H bond, making CF₃OH a much stronger acid than CH₃OH.
2. Inductive Effect: The Distance Matters
The inductive effect describes the transmission of charge through a molecule via sigma bonds. Electron-withdrawing groups (EWGs), like halogens (F, Cl, Br, I), nitro groups (NO₂), and cyano groups (CN), stabilize the conjugate base by pulling electron density away from the negatively charged atom. This stabilization makes it easier to donate the proton, increasing acidity. The strength of the inductive effect diminishes with distance, meaning EWGs closer to the acidic proton have a greater effect.
Example: Consider acetic acid (CH₃COOH) and chloroacetic acid (ClCH₂COOH). The chlorine atom in chloroacetic acid is an EWG, exerting an inductive effect that stabilizes the conjugate base (acetate ion). This makes chloroacetic acid a stronger acid than acetic acid.
3. Resonance: Sharing the Burden
Resonance occurs when electrons can be delocalized over multiple atoms, creating multiple equivalent structures. If the conjugate base can participate in resonance, the negative charge is spread out over a larger area, significantly stabilizing the anion and increasing the acidity of the parent acid.
Example: Compare phenol (C₆H₅OH) and cyclohexanol (C₆H₁₁OH). The phenoxide ion (conjugate base of phenol) can participate in resonance, delocalizing the negative charge over the aromatic ring. This stabilization makes phenol significantly more acidic than cyclohexanol, where no resonance stabilization is possible.
4. Hybridization: Orbital Influence
The hybridization of the atom bonded to the acidic proton influences acidity. Atoms with higher s-character (sp > sp² > sp³) hold electrons more tightly, resulting in a stronger bond and decreased acidity. Conversely, atoms with lower s-character hold electrons less tightly, resulting in weaker bonds and increased acidity.
Example: Acetylene (sp hybridized carbon) is less acidic than ethylene (sp² hybridized carbon), which is less acidic than ethane (sp³ hybridized carbon). The increasing s-character leads to stronger C-H bonds and reduced acidity.
5. Size and Polarizability: The Larger, the Better
In some cases, the size and polarizability of an atom influence acidity. Larger atoms with more diffuse electron clouds are better able to stabilize the negative charge in the conjugate base through polarizability. This effect is particularly prominent when comparing acids with larger halogens or heavier chalcogens.
Example: HI is a stronger acid than HF. The larger size of iodine allows for better dispersion of the negative charge in the iodide ion (I⁻), making it more stable compared to the fluoride ion (F⁻).
Arranging Compounds: A Step-by-Step Approach
To arrange compounds in order of increasing acidity, systematically consider the factors mentioned above. Here's a structured approach:
-
Identify the acidic proton: Determine which hydrogen atom is most likely to be donated as a proton (H⁺). This is usually associated with an oxygen or nitrogen atom, but could also involve a carbon atom in specific cases (like terminal alkynes).
-
Analyze the conjugate base: Consider the stability of the conjugate base that forms after the proton is donated. A more stable conjugate base implies a stronger acid.
-
Evaluate electronegativity: Identify the presence and proximity of electronegative atoms. The more electronegative atoms near the acidic proton, the stronger the acid.
-
Assess resonance effects: Determine if the conjugate base can participate in resonance. Resonance stabilization significantly increases acidity.
-
Consider inductive effects: Identify electron-withdrawing or electron-donating groups and their proximity to the acidic proton. Electron-withdrawing groups enhance acidity, while electron-donating groups reduce it.
-
Evaluate hybridization: Consider the hybridization of the atom bonded to the acidic proton. Higher s-character leads to lower acidity.
-
Compare sizes and polarizabilities: In cases of similar structures, consider the size and polarizability of atoms involved. Larger, more polarizable atoms stabilize the negative charge better, leading to higher acidity.
Examples and Practice Problems
Let's apply this approach to some examples:
Example 1: Arrange the following compounds in order of increasing acidity: CH₃OH, CF₃COOH, CH₃COOH, H₂O.
- CH₃OH: Relatively weak acid, only weakly stabilized conjugate base.
- H₂O: Slightly stronger than methanol, but still weak.
- CH₃COOH: Stronger than water and methanol due to resonance stabilization of the conjugate base (acetate ion).
- CF₃COOH: Strongest acid due to the strong electron-withdrawing effect of three fluorine atoms.
Order: CH₃OH < H₂O < CH₃COOH < CF₃COOH
Example 2: Arrange the following compounds in order of increasing acidity: phenol, ethanol, acetic acid.
- Ethanol: Weak acid, no resonance stabilization of the conjugate base.
- Phenol: Moderately acidic due to resonance stabilization of the phenoxide ion.
- Acetic acid: Stronger than both ethanol and phenol, due to both resonance stabilization and the inductive effect of the carbonyl group.
Order: Ethanol < Phenol < Acetic acid
Example 3: A More Complex Scenario Arrange the following compounds in order of increasing acidity: HCl, HBr, HI, HF.
All are strong acids, but their acidity varies due to differences in bond strength and stability of the conjugate bases.
- HF: The strongest bond, least stable conjugate base due to high electronegativity and small size of Fluorine. This results in lower acidity than the others.
- HCl: Stronger than HF, with a weaker bond and a more stable conjugate base.
- HBr: Stronger than HCl, due to a further increase in bond length and stability of the bromide anion.
- HI: The strongest acid because of the weakest bond and the largest and most stable iodide anion.
Order: HF < HCl < HBr < HI
By carefully analyzing each factor for every given compound, one can systematically arrange compounds in the correct order of increasing acidity. Remember to meticulously examine the structure and properties of each molecule, focusing on conjugate base stability as the central determining element. Practice is key to mastering this essential chemistry skill. The more examples you work through, the more intuitive the process will become. Remember to always consider the interplay of all contributing factors for a comprehensive understanding.
Latest Posts
Latest Posts
-
Which Of The Following Is A Disadvantage Of Franchising
Apr 06, 2025
-
Which Of The Following Does Not Occur After Menopause
Apr 06, 2025
-
Independent Variable Dependent Variable Control Variable
Apr 06, 2025
-
A Joint Between Bones Of The Skull Is A
Apr 06, 2025
-
Number Of Valence Electrons In Ca
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Arrange The Compounds In Order Of Increasing Acidity. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
