Are Metals Solid At Room Temperature
News Leon
Apr 02, 2025 · 5 min read
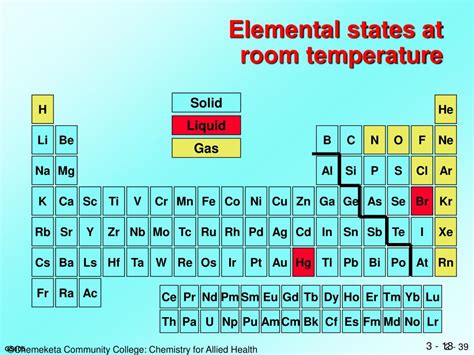
Table of Contents
Are Metals Solid at Room Temperature? Exploring the Exceptions and the Rules
The simple answer is yes, most metals are solid at room temperature. This is a fundamental property that underpins their widespread use in countless applications, from construction to electronics. However, the seemingly straightforward statement hides a fascinating complexity. This article delves deep into the world of metals, exploring why they are typically solid at room temperature, examining the exceptions to this rule, and considering the factors that influence their physical state.
The Atomic Structure: A Foundation for Solidity
The solidity of most metals at room temperature is directly linked to their atomic structure and the nature of metallic bonding. Unlike other types of bonding, metallic bonding involves a "sea" of delocalized electrons that are not associated with any particular atom. These electrons are free to move throughout the metal lattice, creating a strong electrostatic attraction between the positively charged metal ions and the negatively charged electron cloud.
The Role of Metallic Bonding
This unique bonding mechanism is responsible for several key properties of metals, including their:
-
High melting and boiling points: The strong electrostatic attraction requires significant energy to overcome, resulting in high melting and boiling points. This is why most metals remain solid at room temperature (approximately 25°C or 77°F).
-
Malleability and ductility: The delocalized electrons allow the metal ions to slide past each other without disrupting the overall structure, making metals malleable (capable of being hammered into shapes) and ductile (capable of being drawn into wires).
-
Electrical and thermal conductivity: The free-moving electrons are also responsible for the excellent electrical and thermal conductivity of metals. Heat and electricity can easily flow through the metal lattice due to the mobility of these electrons.
-
Luster: The interaction of light with the delocalized electrons gives metals their characteristic luster or shine.
Exceptions to the Rule: Mercury and Others
While the vast majority of metals are solid at room temperature, there are notable exceptions. The most prominent example is mercury (Hg), a liquid at room temperature. This unusual behavior stems from several factors:
-
Weak metallic bonding: Although mercury exhibits metallic bonding, the interaction between its atoms is relatively weak compared to other metals. This weak bonding is partly due to the relativistic effects on its 6s electrons, which are unusually tightly bound to the nucleus.
-
Relativistic Effects: The high atomic number of mercury leads to relativistic effects on its electrons. These effects influence the size of the electron orbitals and impact the strength of metallic bonds. This is a complex quantum mechanical phenomenon, but essentially, the relativistic contraction of the 6s orbitals reduces the effectiveness of the metallic bonding.
-
Liquid State at Room Temperature: The combination of weak metallic bonding and relativistic effects results in mercury's low melting point (-38.83 °C or -37.89 °F), making it a liquid at room temperature. This is a unique property amongst the common metals and has made it useful in various applications throughout history, although its toxicity has significantly curtailed its use.
Beyond mercury, some other metals possess melting points close to room temperature. These include:
-
Cesium (Cs): Melting point of 28.44 °C (83.19 °F). Cesium is a highly reactive alkali metal and must be stored under oil to prevent oxidation.
-
Gallium (Ga): Melting point of 29.76 °C (85.57 °F). Gallium's low melting point makes it useful in various low-temperature applications and has a notable characteristic of melting in the hand.
-
Francium (Fr): A radioactive alkali metal with an extremely low melting point, predicted to be even lower than cesium. Its extreme radioactivity prevents detailed study of its physical properties.
These metals, while exceptions, still exhibit metallic bonding. Their lower melting points simply reflect weaker interatomic forces compared to most other metals.
Factors Influencing the Melting Point of Metals
Several factors contribute to the variation in melting points among different metals:
-
Atomic size and mass: Larger atoms generally have weaker metallic bonds due to greater atomic radii and thus lower melting points.
-
Number of valence electrons: The number of valence electrons available for metallic bonding influences the strength of the bond. Metals with more valence electrons tend to have stronger bonds and higher melting points.
-
Crystal structure: The arrangement of atoms in the metal lattice can also affect the strength of metallic bonds. Different crystal structures (e.g., body-centered cubic, face-centered cubic) can lead to variations in melting points.
-
Alloys and Impurities: The presence of alloying elements or impurities can significantly affect the melting point of a metal. Alloying is a common technique used to alter the properties of metals, including their melting points.
-
Pressure: Pressure can also influence the melting point of a metal. Increased pressure generally increases the melting point.
Practical Applications and Implications
The fact that most metals are solid at room temperature is crucial for their use in numerous applications:
-
Construction: Steel, iron, aluminum, and other metals form the backbone of buildings, bridges, and other structures. Their solidity and strength are essential for their structural integrity.
-
Manufacturing: Metals are extensively used in the manufacturing of machinery, tools, and various other products. Their ability to maintain their shape at room temperature is vital for manufacturing processes.
-
Transportation: Cars, airplanes, trains, and ships all rely heavily on metallic components. The strength and durability of metals are crucial for their safety and functionality.
-
Electronics: Metals play a vital role in electronic devices, serving as conductors in circuits and components. Their electrical conductivity is essential for the operation of these devices.
Conclusion: A Solid Foundation
In summary, while there are exceptions, the vast majority of metals are indeed solid at room temperature due to the strong metallic bonding between their atoms. This property, combined with other characteristics like malleability, ductility, and conductivity, accounts for their wide range of applications and their importance in modern society. Understanding the atomic structure, bonding, and factors that influence melting points provides a comprehensive picture of why metals, in their solid form, are such fundamental materials. The exceptions, like mercury, highlight the intricate interplay of various physical and chemical forces that dictate the properties of matter. Further research into the behavior of metals under extreme conditions, such as high temperatures and pressures, continues to expand our understanding of this vital class of materials.
Latest Posts
Latest Posts
-
How Many Watts Equal One Horse Power
Apr 03, 2025
-
Which Element Has The Lowest First Ionization Energy
Apr 03, 2025
-
Which Of The Following Is Not Found In Blood Plasma
Apr 03, 2025
-
Compare The Pectoral And Pelvic Girdles
Apr 03, 2025
-
Find The Values Of X And Y In Parallelogram Pqrs
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Are Metals Solid At Room Temperature . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
