Which Element Has The Lowest First Ionization Energy
News Leon
Apr 03, 2025 · 5 min read
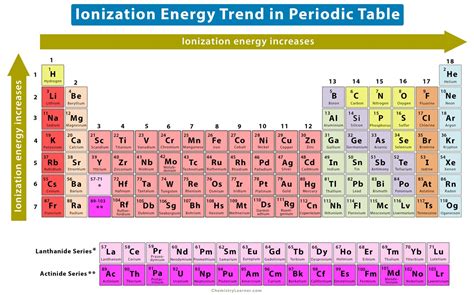
Table of Contents
Which Element Has the Lowest First Ionization Energy? Understanding Ionization Energy Trends
The quest to identify the element with the lowest first ionization energy involves delving into the fascinating world of atomic structure and periodic trends. Understanding ionization energy, its variations across the periodic table, and the factors influencing it is crucial for comprehending chemical reactivity and behavior. This comprehensive article will explore these aspects, ultimately revealing the element with the lowest first ionization energy and explaining why.
What is Ionization Energy?
Ionization energy is the minimum amount of energy required to remove the most loosely bound electron from a neutral gaseous atom or ion. This process transforms a neutral atom into a positively charged ion (cation). The first ionization energy specifically refers to the energy needed to remove the first electron. Subsequent ionization energies (second, third, etc.) refer to the removal of subsequent electrons, each requiring progressively more energy.
The ionization energy is typically expressed in kilojoules per mole (kJ/mol) or electron volts (eV). A lower ionization energy signifies that it's easier to remove an electron, indicating a less tightly bound electron. Conversely, a higher ionization energy implies a more tightly bound electron, requiring more energy for removal.
Factors Affecting Ionization Energy
Several factors significantly influence an element's ionization energy:
-
Effective Nuclear Charge (Z<sub>eff</sub>): This is the net positive charge experienced by an electron in an atom. A higher Z<sub>eff</sub> means the electrons are more strongly attracted to the nucleus, resulting in a higher ionization energy. Shielding by inner electrons reduces Z<sub>eff</sub>.
-
Atomic Radius: A larger atomic radius implies a greater distance between the nucleus and the outermost electrons. This increased distance weakens the electrostatic attraction between the nucleus and the electrons, leading to a lower ionization energy.
-
Electron Shielding: Inner electrons shield the outer electrons from the full positive charge of the nucleus. Greater shielding reduces the effective nuclear charge experienced by outer electrons, lowering the ionization energy.
-
Electron Configuration: The specific arrangement of electrons in an atom influences its ionization energy. Electrons in filled subshells (e.g., s<sup>2</sup>, p<sup>6</sup>) are generally more stable and require more energy to remove than electrons in partially filled subshells.
Periodic Trends in Ionization Energy
Ionization energy exhibits predictable trends across the periodic table:
-
Across a Period (Left to Right): Ionization energy generally increases as you move from left to right across a period. This is primarily due to the increasing effective nuclear charge. As you add protons and electrons, the increased positive charge pulls the electrons closer, making them harder to remove.
-
Down a Group (Top to Bottom): Ionization energy generally decreases as you move down a group. This is attributed to the increasing atomic radius and increased shielding by inner electrons. The larger distance between the nucleus and the outermost electrons weakens the attraction, making it easier to remove an electron.
Identifying the Element with the Lowest First Ionization Energy
Based on the periodic trends discussed above, elements located at the bottom left of the periodic table will have the lowest first ionization energy. This is because they possess:
- Large atomic radii: The outermost electrons are far from the nucleus, experiencing weak attraction.
- Low effective nuclear charge: The shielding effect of inner electrons significantly reduces the positive charge felt by the valence electrons.
- One loosely held valence electron: The tendency is to readily lose this electron to achieve a stable electron configuration.
Considering these factors, the element with the lowest first ionization energy is Francium (Fr).
Why Francium?
Francium is an alkali metal located in the bottom-left corner of the periodic table. It has the largest atomic radius among all naturally occurring elements. This large distance between its nucleus and its single valence electron results in extremely weak electrostatic attraction. The high number of electron shells and efficient shielding further reduces the effective nuclear charge, making it exceptionally easy to remove the single valence electron.
It's important to note that Francium is extremely rare and radioactive, making experimental determination of its ionization energy challenging. However, based on established periodic trends and theoretical calculations, it's undeniably the element with the lowest first ionization energy.
Comparison with Other Alkali Metals
While Francium holds the title, let's briefly compare it with other alkali metals:
-
Cesium (Cs): Immediately above Francium, Cesium also has a very low first ionization energy. It has a smaller atomic radius than Francium but still exhibits a relatively weak hold on its valence electron.
-
Rubidium (Rb), Potassium (K), Sodium (Na), Lithium (Li): As we move up Group 1, the atomic radius decreases, and the ionization energy increases accordingly. Lithium, at the top of the group, has a significantly higher first ionization energy than Francium or Cesium.
Applications of Ionization Energy
Understanding ionization energy is crucial in various fields:
-
Chemistry: It helps predict chemical reactivity. Elements with low ionization energies readily lose electrons and form cations, while those with high ionization energies tend to gain electrons and form anions.
-
Physics: Ionization energies are essential in understanding atomic and molecular structure, spectroscopy, and plasma physics.
-
Material Science: Ionization energy influences the properties of materials, impacting their conductivity, reactivity, and other physical characteristics.
Conclusion: Francium Reigns Supreme
In conclusion, Francium (Fr) unequivocally holds the title of the element with the lowest first ionization energy. Its unique position in the periodic table, characterized by its extremely large atomic radius, low effective nuclear charge, and a single loosely held valence electron, makes it exceptionally prone to losing an electron. While its rarity and radioactivity hinder direct experimental verification, theoretical predictions and established periodic trends firmly support its claim as the champion of low ionization energy. Understanding this fundamental property is key to comprehending the behavior and reactivity of elements within the broader context of chemistry and physics. This knowledge serves as a cornerstone in many scientific disciplines, furthering our understanding of the atomic world and its impact on the macroscopic properties of matter.
Latest Posts
Latest Posts
-
Which Of The Following Is A Non Renewable Source Of Energy
Apr 04, 2025
-
What Binds To The Exposed Cross Bridges On Actin
Apr 04, 2025
-
All Squares Are Rectangles And Rhombuses
Apr 04, 2025
-
What Is The Role Of Toothpaste In Preventing Cavities
Apr 04, 2025
-
Is A Grasshopper A Producer Consumer Or Decomposer
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Element Has The Lowest First Ionization Energy . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
