Which Of The Following Alcohols Dehydrates With The Fastest Rate
News Leon
Apr 03, 2025 · 5 min read
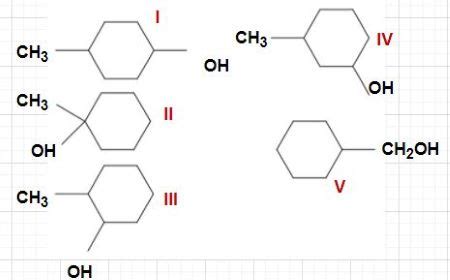
Table of Contents
Which Alcohol Dehydrates Fastest? A Deep Dive into Dehydration Kinetics
The dehydration of alcohols, a fundamental reaction in organic chemistry, involves the removal of a water molecule from an alcohol to form an alkene. However, not all alcohols dehydrate at the same rate. The speed of this reaction is significantly influenced by several factors, including the structure of the alcohol itself. This article will delve into the kinetics of alcohol dehydration, exploring the factors that govern reaction rate and ultimately answering the question: which alcohol dehydrates fastest?
Factors Affecting Alcohol Dehydration Rate
Several key factors contribute to the variation in dehydration rates among different alcohols:
1. The Nature of the Alcohol: Primary, Secondary, and Tertiary Alcohols
The type of alcohol (primary, secondary, or tertiary) drastically impacts the dehydration rate. This difference arises from the stability of the carbocation intermediate formed during the reaction. The mechanism generally involves the protonation of the hydroxyl group, followed by the departure of a water molecule, creating a carbocation. This carbocation is then deprotonated to yield the alkene.
-
Tertiary alcohols: These alcohols dehydrate the fastest. The reason lies in the stability of the tertiary carbocation. Tertiary carbocations are highly stable due to the presence of three alkyl groups that donate electron density, stabilizing the positive charge. This stability significantly lowers the activation energy of the reaction, leading to a rapid dehydration.
-
Secondary alcohols: These dehydrate at a moderate rate. The secondary carbocation intermediate is less stable than a tertiary carbocation but more stable than a primary carbocation. Consequently, the dehydration rate is slower than tertiary alcohols but faster than primary alcohols.
-
Primary alcohols: These dehydrate at the slowest rate. The primary carbocation intermediate is the least stable among the three types. Its instability leads to a high activation energy, resulting in a significantly slower dehydration rate. In fact, primary alcohols often require more vigorous conditions (higher temperatures and/or stronger acids) to undergo dehydration.
2. Steric Hindrance
The presence of bulky groups around the hydroxyl group can hinder the approach of the acid catalyst and the subsequent formation of the carbocation. This steric hindrance slows down the dehydration reaction. For example, a tertiary alcohol with bulky substituents might dehydrate slower than a tertiary alcohol with smaller substituents, even though both are tertiary alcohols.
3. The Strength of the Acid Catalyst
The strength of the acid catalyst employed also influences the reaction rate. Stronger acids like sulfuric acid (H₂SO₄) or phosphoric acid (H₃PO₄) protonate the alcohol more readily, increasing the rate of dehydration. Weaker acids will lead to a slower reaction rate.
4. Reaction Temperature
The temperature significantly affects the rate of dehydration. Higher temperatures provide the molecules with more kinetic energy, increasing the likelihood of successful collisions leading to the formation of the carbocation and subsequent alkene. Thus, higher temperatures accelerate the dehydration reaction.
Comparing Dehydration Rates: Specific Examples
Let's consider some specific examples to illustrate the differences in dehydration rates:
Scenario 1: Comparing Tertiary, Secondary, and Primary Alcohols with similar structures.
Imagine comparing the dehydration of the following alcohols:
- 2-Methyl-2-propanol (tert-butyl alcohol): A tertiary alcohol.
- 2-Butanol (sec-butyl alcohol): A secondary alcohol.
- 1-Butanol (n-butyl alcohol): A primary alcohol.
Under identical reaction conditions (same acid catalyst, temperature, concentration), 2-methyl-2-propanol will dehydrate the fastest, followed by 2-butanol, and then 1-butanol. This is a direct consequence of the relative stability of the carbocations formed during the reaction.
Scenario 2: The Influence of Steric Hindrance.
Consider the dehydration of two tertiary alcohols:
- 2-Methyl-2-propanol (tert-butyl alcohol): Relatively small substituents.
- 2,2-Dimethyl-3-hexanol: Larger, bulkier substituents.
Even though both are tertiary alcohols, 2-methyl-2-propanol is likely to dehydrate faster due to less steric hindrance around the hydroxyl group. The bulky substituents in 2,2-dimethyl-3-hexanol hinder the approach of the acid catalyst and the formation of the carbocation, thus slowing down the reaction.
Scenario 3: The Effect of Acid Catalyst Strength.
If we dehydrate the same alcohol (e.g., 2-butanol) using different acids, the stronger acid will lead to a faster reaction rate. For instance, using concentrated sulfuric acid will result in a faster dehydration than using a dilute solution of phosphoric acid.
Practical Implications and Applications
Understanding the factors that influence the dehydration rate of alcohols has significant practical implications in various fields:
-
Organic Synthesis: Chemists carefully select reaction conditions (type of alcohol, acid catalyst, temperature) to control the rate and yield of alkene formation during organic synthesis. The ability to manipulate the dehydration rate is crucial for achieving desired selectivity and efficiency in various chemical processes.
-
Industrial Processes: The dehydration of alcohols is used in numerous industrial processes for the production of various alkenes. Optimization of dehydration rates is vital for improving the efficiency and cost-effectiveness of these industrial processes.
Conclusion: Speed Isn't Everything, but it Matters
While the specific rate of dehydration can vary based on numerous experimental factors, the general trend is clear: tertiary alcohols generally dehydrate the fastest, followed by secondary alcohols, and then primary alcohols. However, steric hindrance, the strength of the acid catalyst, and the reaction temperature all play crucial roles in modulating the overall reaction rate. Understanding these factors is essential for predicting and controlling the outcomes of alcohol dehydration reactions in various contexts, from academic research to industrial applications. The interplay of these factors highlights the complexity and elegance of organic chemistry reactions.
Latest Posts
Latest Posts
-
Containing Two Different Alleles For A Trait
Apr 04, 2025
-
Which Of The Following Is A Non Renewable Source Of Energy
Apr 04, 2025
-
What Binds To The Exposed Cross Bridges On Actin
Apr 04, 2025
-
All Squares Are Rectangles And Rhombuses
Apr 04, 2025
-
What Is The Role Of Toothpaste In Preventing Cavities
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Alcohols Dehydrates With The Fastest Rate . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
