Which Compound Is A Secondary Amine
News Leon
Apr 05, 2025 · 6 min read
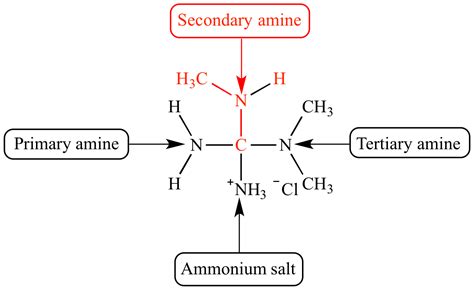
Table of Contents
Which Compound is a Secondary Amine? A Deep Dive into Amine Classification
Amines are organic compounds derived from ammonia (NH₃) by replacing one or more hydrogen atoms with alkyl or aryl groups. Understanding their classification, particularly differentiating between primary, secondary, and tertiary amines, is crucial in organic chemistry. This article will provide a comprehensive guide to identifying secondary amines, exploring their properties, and examining examples. We'll delve into the structural features that define them and discuss their importance in various applications.
Understanding Amine Classification: Primary, Secondary, and Tertiary
The classification of amines hinges on the number of alkyl or aryl groups attached to the nitrogen atom. This seemingly simple distinction significantly impacts their chemical behavior and reactivity.
Primary Amines (1° Amines):
A primary amine has one alkyl or aryl group attached to the nitrogen atom. The general formula is R-NH₂, where R represents the alkyl or aryl group. Examples include methylamine (CH₃NH₂) and aniline (C₆H₅NH₂).
Secondary Amines (2° Amines):
A secondary amine features two alkyl or aryl groups bonded to the nitrogen atom. Its general formula is R₁-NH-R₂, where R₁ and R₂ can be the same or different alkyl or aryl groups. This is the focus of our detailed exploration.
Tertiary Amines (3° Amines):
Tertiary amines possess three alkyl or aryl groups attached to the nitrogen atom. Their general formula is R₁-N-R₂-R₃, where R₁, R₂, and R₃ can be the same or different alkyl or aryl groups.
Identifying Secondary Amines: Key Structural Features
The defining characteristic of a secondary amine is the presence of a nitrogen atom bonded to exactly two carbon atoms (or one carbon and one other group such as an aryl group). This nitrogen atom also has one lone pair of electrons, which contributes to their basicity and reactivity.
Let's visualize this:
R₁
|
N - R₂
|
H
In this structure:
- N: Represents the nitrogen atom.
- R₁ and R₂: Represent the alkyl or aryl groups attached to the nitrogen. These groups can be identical (e.g., dimethylamine) or different (e.g., ethylmethylamine).
- H: Represents a single hydrogen atom attached to the nitrogen. This is a crucial distinction from tertiary amines.
Examples of Secondary Amines: A Diverse Range of Compounds
Secondary amines are found in a vast array of compounds, both naturally occurring and synthetic. Let's look at some key examples:
Simple Alkyl Secondary Amines:
- Dimethylamine (CH₃-NH-CH₃): A simple secondary amine with two methyl groups attached to the nitrogen. It's a colorless gas with a fishy odor and is used in the production of various chemicals.
- Diethylamine (CH₃CH₂-NH-CH₂CH₃): Similar to dimethylamine, but with two ethyl groups. It's a liquid with a characteristic ammonia-like odor and is employed as a solvent and in the synthesis of other compounds.
- Ethylmethylamine (CH₃-NH-CH₂CH₃): This illustrates a secondary amine with two different alkyl groups, methyl and ethyl, attached to the nitrogen.
Cyclic Secondary Amines:
- Pyrrolidine: A five-membered heterocyclic ring containing a nitrogen atom. The nitrogen atom is bonded to two carbon atoms within the ring, making it a secondary amine. Pyrrolidine is found in various alkaloids and is used in organic synthesis.
- Piperidine: A six-membered heterocyclic ring containing a nitrogen atom. Similar to pyrrolidine, the nitrogen atom is bonded to two carbon atoms within the ring and is thus a secondary amine. Piperidine is also found in several natural products and used in chemical synthesis.
- Morpholine: A six-membered heterocyclic ring with both oxygen and nitrogen atoms. The nitrogen atom is bonded to two carbon atoms, one of which is part of an ether linkage, making it a secondary amine. Morpholine is used as a solvent and in the synthesis of pharmaceuticals.
Aryl-Alkyl Secondary Amines:
- N-methylaniline (C₆H₅-NH-CH₃): This amine contains a phenyl group (aryl) and a methyl group (alkyl) attached to the nitrogen. It's an important intermediate in the synthesis of dyes and pharmaceuticals.
- N-ethylaniline (C₆H₅-NH-CH₂CH₃): Similar to N-methylaniline, but with an ethyl group instead of a methyl group.
Distinguishing Secondary Amines from Other Amine Types
It's essential to be able to differentiate secondary amines from primary and tertiary amines. Several methods can be employed:
1. Spectroscopic Techniques:
- NMR Spectroscopy (Nuclear Magnetic Resonance): ¹H NMR can identify the characteristic chemical shift of the proton attached to the nitrogen in secondary amines. The chemical shift of the N-H proton is often broad and can vary depending on the solvent and concentration. The signals of the alkyl or aryl groups attached to the nitrogen also provide valuable structural information.
- IR Spectroscopy (Infrared Spectroscopy): IR spectroscopy can detect the N-H stretching vibration in secondary amines, typically appearing in the range of 3300-3100 cm⁻¹. This peak is usually less sharp than the N-H stretching peak in primary amines.
2. Chemical Tests:
Several chemical tests can be used to distinguish between different types of amines. While not definitive for identifying secondary amines specifically, these tests can help narrow down the possibilities based on the reactions observed. These tests often rely on the reactivity of the lone pair of electrons on the nitrogen atom.
Applications of Secondary Amines: A Wide Range of Uses
Secondary amines play vital roles in various applications due to their unique chemical properties.
1. Pharmaceuticals:
Many pharmaceuticals contain secondary amine functionalities within their structures. These amines may contribute to the drug's activity, bioavailability, or stability. Many drugs containing secondary amines target specific receptors or enzymes within the body.
2. Polymers:
Secondary amines are used in the synthesis of various polymers, such as polyamides (nylons) and polyurethanes. The amine group participates in the polymerization reaction, contributing to the properties of the resulting polymer.
3. Solvents:
Certain secondary amines act as solvents in various industrial and laboratory applications. Their ability to dissolve a wide range of compounds makes them valuable in chemical processes.
4. Catalysts:
Secondary amines are sometimes used as catalysts in various chemical reactions. Their ability to act as both a base and a nucleophile allows them to participate in diverse catalytic cycles.
5. Pesticides and Herbicides:
Some secondary amines are incorporated into pesticides and herbicides to enhance their effectiveness or target specific pests or weeds. The amine functionality may contribute to the compound's binding affinity to the target organism.
6. Dyes and Pigments:
Certain secondary amines are incorporated into the structures of dyes and pigments, influencing their color and properties. The amine group may participate in the formation of chromophores or contribute to the dye's interaction with the substrate.
Conclusion: A Critical Functional Group in Organic Chemistry
Secondary amines represent a significant class of organic compounds with diverse structures and applications. Their unique chemical properties stem from the presence of the nitrogen atom bonded to two alkyl or aryl groups and a single hydrogen atom. Understanding their structure, properties, and synthesis is crucial in various fields, including medicinal chemistry, materials science, and industrial chemistry. This comprehensive exploration has highlighted the importance of correctly identifying secondary amines through various techniques and understanding their widespread use in numerous applications. Further exploration into the reactivity and specific applications of individual secondary amines will provide a deeper understanding of their significance in organic chemistry.
Latest Posts
Latest Posts
-
How Many Meters Are In 600 Centimeters
Apr 05, 2025
-
Factor X 4 5x 2 4
Apr 05, 2025
-
How Much Atp Is Produced In Fermentation
Apr 05, 2025
-
Is Condensation Of Steam A Chemical Change
Apr 05, 2025
-
A Rectangle Is A Parallelogram With A Right Interior Angle
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Which Compound Is A Secondary Amine . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
