When Dissolved In Water Acids Produce
News Leon
Apr 03, 2025 · 6 min read
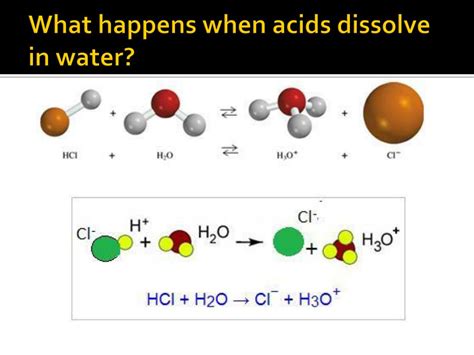
Table of Contents
When Dissolved in Water, Acids Produce: A Deep Dive into Acid-Base Chemistry
Acids are ubiquitous in our lives, from the citric acid in oranges to the sulfuric acid used in industrial processes. Understanding their behavior, particularly when dissolved in water, is crucial for numerous scientific fields and everyday applications. This comprehensive article will explore the fundamental processes that occur when acids dissolve in water, focusing on the resulting ions and their implications. We'll examine different acid strengths, explore the concept of pH, and discuss the practical applications of this fundamental chemical reaction.
The Nature of Acids: A Brief Overview
Before delving into the specifics of what happens when acids dissolve in water, let's establish a basic understanding of acids themselves. Acids are substances that donate protons (H⁺ ions) when dissolved in a solution. This is according to the Brønsted-Lowry definition of acids, a widely accepted model in chemistry. Another definition, the Arrhenius definition, focuses on acids increasing the concentration of hydrogen ions (H⁺) in aqueous solutions. While simpler, the Brønsted-Lowry definition provides a broader perspective, encompassing reactions that don't necessarily involve water.
Acids exhibit certain characteristic properties, including:
- Sour taste: This is a classic indicator, though it should never be tested directly with unknown substances due to potential hazards.
- Reaction with metals: Many acids react with active metals (like zinc or magnesium) to produce hydrogen gas.
- Change in litmus paper color: Acids turn blue litmus paper red.
- Reaction with bases: Acids neutralize bases, forming salt and water in a process known as neutralization.
What Happens When Acids Dissolve in Water? The Production of Hydronium Ions
The key event when an acid dissolves in water is the donation of a proton (H⁺) to a water molecule (H₂O). This process results in the formation of a hydronium ion (H₃O⁺). It's crucial to understand that free protons (H⁺) don't exist independently in aqueous solutions; they immediately bond with a water molecule.
Let's illustrate this with the example of hydrochloric acid (HCl), a strong acid:
HCl(aq) + H₂O(l) → H₃O⁺(aq) + Cl⁻(aq)
In this reaction:
- HCl, a strong acid, readily donates its proton (H⁺).
- H₂O acts as a proton acceptor, or a Brønsted-Lowry base.
- H₃O⁺, the hydronium ion, is formed.
- Cl⁻, the chloride ion, is the conjugate base of HCl. The conjugate base is the remaining part of the acid after it has donated a proton.
This reaction highlights the essential characteristic of acids in aqueous solutions: the production of hydronium ions. The concentration of hydronium ions directly determines the acidity of the solution.
Strong Acids vs. Weak Acids: A Key Distinction
The extent to which an acid dissociates (donates protons) in water dictates its strength. Strong acids completely dissociate into ions when dissolved in water, meaning virtually all the acid molecules donate their protons. Examples include:
- Hydrochloric acid (HCl)
- Sulfuric acid (H₂SO₄)
- Nitric acid (HNO₃)
- Perchloric acid (HClO₄)
- Hydrobromic acid (HBr)
- Hydroiodic acid (HI)
Weak acids, on the other hand, only partially dissociate in water. A significant portion of the acid molecules remain undissociated, existing in equilibrium with their ions. Examples include:
- Acetic acid (CH₃COOH) (found in vinegar)
- Carbonic acid (H₂CO₃) (found in carbonated drinks)
- Phosphoric acid (H₃PO₄)
- Hydrofluoric acid (HF)
- Benzoic acid (C₇H₆O₂)
The difference in dissociation is crucial. Strong acids lead to higher hydronium ion concentrations than weak acids at the same molar concentration. This difference is reflected in their pH values.
The pH Scale: Quantifying Acidity
The pH scale is a logarithmic scale used to express the acidity or basicity of a solution. It ranges from 0 to 14, with:
- pH 7: Neutral (equal concentrations of H₃O⁺ and OH⁻ ions)
- pH < 7: Acidic (higher concentration of H₃O⁺ ions)
- pH > 7: Basic or alkaline (higher concentration of OH⁻ ions)
The pH is calculated using the following formula:
pH = -log₁₀[H₃O⁺]
where [H₃O⁺] represents the concentration of hydronium ions in moles per liter (M). A lower pH indicates a higher concentration of hydronium ions and thus greater acidity. For example, a solution with a pH of 1 is ten times more acidic than a solution with a pH of 2.
The Impact of Hydronium Ions: Practical Applications
The production of hydronium ions when acids dissolve in water has far-reaching consequences across various applications:
1. Industrial Processes:
- Chemical synthesis: Acids are essential catalysts and reactants in numerous industrial chemical processes, including the production of fertilizers, plastics, and pharmaceuticals. The hydronium ions play a critical role in these reactions.
- Metal processing: Acids are used to etch, clean, and dissolve metals. The interaction of hydronium ions with metal surfaces is central to these processes.
- Petroleum refining: Acid-catalyzed reactions are crucial in petroleum refining to convert crude oil into various fuels and other products.
2. Biological Systems:
- Digestion: Stomach acid (primarily hydrochloric acid) provides the low pH environment necessary for the digestion of food. The hydronium ions activate digestive enzymes and break down food molecules.
- Enzyme activity: Many enzymes require a specific pH range to function optimally. The hydronium ion concentration significantly impacts enzyme activity in biological systems.
- Regulation of blood pH: The body maintains a tightly regulated blood pH within a narrow range. Buffers in the blood help to minimize changes in pH caused by the intake of acids or bases.
3. Everyday Life:
- Food and beverages: Many foods and beverages contain acids that contribute to their taste and preservation. The hydronium ions from these acids affect the flavor and shelf life of the products.
- Cleaning: Many household cleaners contain acids that help to remove stains, disinfect surfaces, and dissolve mineral deposits. The acidic nature facilitates these cleaning actions.
- Battery acid: Car batteries use sulfuric acid as an electrolyte. The hydronium ions contribute to the electrochemical reactions that generate electricity.
Beyond Hydronium Ions: Other Aspects of Acid Dissolution
While the formation of hydronium ions is the central event when acids dissolve in water, other aspects are worth considering:
- Anion behavior: The conjugate base formed (e.g., Cl⁻ from HCl) also plays a role. Some anions can react further with water, influencing the overall pH.
- Concentration effects: The concentration of the acid significantly impacts the hydronium ion concentration and thus the pH. Higher concentrations generally lead to lower pH values.
- Temperature effects: Temperature can affect the rate of acid dissociation and thus the concentration of hydronium ions.
Conclusion: The Significance of Hydronium Ions
When acids dissolve in water, they produce hydronium ions (H₃O⁺), a fundamental process with far-reaching implications. The concentration of hydronium ions determines the acidity of the solution, influencing its pH and affecting a wide range of chemical and biological processes. Understanding this fundamental reaction is critical across various scientific disciplines and is essential in numerous industrial, biological, and everyday applications. From the digestive system to industrial chemical synthesis, the role of hydronium ions is undeniable, highlighting the importance of acid-base chemistry in our world.
Latest Posts
Latest Posts
-
Give The Iupac Name Of The Carboxylic Acid Below
Apr 04, 2025
-
An Interior Angle Of A Regular Polygon Measures 170
Apr 04, 2025
-
A Fluids Resistance To Flow Is Called
Apr 04, 2025
-
Match The Location With The Appropriate Epithelial Tissue
Apr 04, 2025
-
Sodium Sulfate Barium Chloride Balanced Equation
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about When Dissolved In Water Acids Produce . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
