Give The Iupac Name Of The Carboxylic Acid Below.
News Leon
Apr 04, 2025 · 5 min read
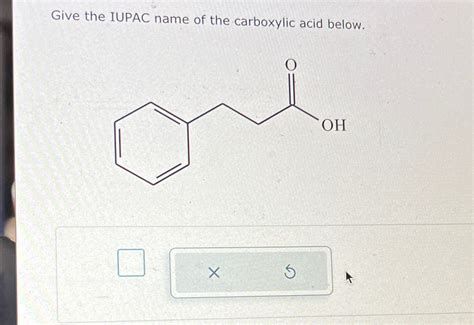
Table of Contents
Give the IUPAC Name of the Carboxylic Acid Below: A Comprehensive Guide
Naming organic compounds, especially carboxylic acids, can seem daunting at first. However, with a systematic approach and a solid understanding of IUPAC nomenclature rules, it becomes a straightforward process. This article will delve deep into naming carboxylic acids, providing you with a comprehensive guide, examples, and tips to master this essential skill in organic chemistry. We'll explore the rules, tackle complex structures, and even touch upon some common pitfalls to avoid. Let's get started!
Understanding the IUPAC System for Carboxylic Acids
The International Union of Pure and Applied Chemistry (IUPAC) provides a standardized system for naming organic compounds, ensuring consistent communication across the global scientific community. For carboxylic acids, this system involves several key steps:
1. Identifying the Parent Chain
The parent chain is the longest continuous carbon chain containing the carboxyl group (-COOH). This is crucial because the entire name is based on this chain. Remember, the carbon atom of the carboxyl group is always counted as part of the parent chain.
2. Numbering the Carbon Chain
The carbon chain is numbered starting from the carboxyl carbon, which is always carbon number 1. This is unlike many other functional groups where the numbering might start from the end closest to a substituent.
3. Identifying and Naming Substituents
Substituents are any atoms or groups attached to the parent chain that are not part of the carboxyl group. These are named and their positions (carbon numbers) are indicated using numbers. If multiple substituents are present, they are listed alphabetically, ignoring prefixes like di- or tri-.
4. Combining the Information
The final IUPAC name is constructed by combining the following information:
- Number and name of substituents: This includes the position (number) and the name of each substituent, listed alphabetically.
- Parent chain name: The name of the parent chain is derived from the number of carbons (e.g., meth-, eth-, prop-, but-, pent-, hex-, etc.). The suffix "-oic acid" is added to indicate the presence of the carboxylic acid functional group.
Examples: Step-by-Step IUPAC Naming
Let's work through a few examples to solidify our understanding.
Example 1: A Simple Carboxylic Acid
Imagine a simple carboxylic acid with three carbons: CH₃CH₂COOH
- Parent Chain: The longest chain containing the carboxyl group has three carbons.
- Numbering: The carboxyl carbon is carbon 1.
- Substituents: There are no substituents.
- Name: The parent chain with three carbons is propane. Adding the carboxylic acid suffix gives propanoic acid.
Example 2: A Carboxylic Acid with a Substituent
Consider this molecule: CH₃CH(Cl)CH₂COOH
- Parent Chain: The longest chain containing the carboxyl group has four carbons.
- Numbering: The carboxyl carbon is carbon 1.
- Substituents: There is one chloro substituent on carbon 2.
- Name: The parent chain with four carbons is butane. The substituent is chloro at position 2. The name is 2-chlorobutanoic acid.
Example 3: A Carboxylic Acid with Multiple Substituents
Let's analyze this structure: CH₃CH(CH₃)CH(Br)COOH
- Parent Chain: The longest chain containing the carboxyl group has four carbons.
- Numbering: The carboxyl carbon is carbon 1.
- Substituents: There is a methyl group (CH₃) on carbon 2 and a bromo group (Br) on carbon 3.
- Name: Alphabetizing the substituents gives bromo before methyl. Therefore, the name is 3-bromo-2-methylbutanoic acid.
Example 4: A More Complex Case with Branching
Let's tackle a more complex molecule:
CH₃
|
CH₃-CH-CH₂-CH(CH₃)-COOH
|
CH₂CH₃
- Parent Chain: The longest chain containing the carboxyl group has six carbons.
- Numbering: The carboxyl carbon is carbon 1.
- Substituents: A methyl group (CH₃) on carbon 2, an ethyl group (CH₂CH₃) on carbon 4, and another methyl group on carbon 4.
- Name: Alphabetizing the substituents, we get 4-ethyl, 2-methyl, 4-methyl. Since we have two methyl groups on carbon 4, we use the prefix "di". The final name is 4-ethyl-2,4-dimethylhexanoic acid.
Dealing with Common Challenges in IUPAC Naming
Several aspects of naming carboxylic acids can present challenges for students. Let's address some common difficulties:
1. Identifying the Longest Carbon Chain
Sometimes the longest carbon chain isn't immediately obvious, especially with branched structures. Carefully examine the molecule and try different pathways to ensure you've found the longest possible chain containing the carboxyl group.
2. Correct Numbering
Always remember that numbering begins at the carboxyl carbon (C=O-OH). Incorrect numbering leads to an incorrect name. Double-check your numbering before proceeding to the final naming step.
3. Alphabetical Ordering of Substituents
This is a crucial step. Ignoring prefixes like "di-" or "tri-" during alphabetization is essential. For instance, "dimethyl" comes before "ethyl," even though "di" comes after "e" alphabetically.
4. Handling Multiple Identical Substituents
When multiple identical substituents are present, use prefixes like "di-", "tri-", "tetra-", etc., to indicate their number. Specify the position of each substituent using numbers.
5. Dealing with Complex Substituents
If a substituent itself is a complex group, treat it as a single entity and name it appropriately, including the necessary locants and prefixes.
Advanced Naming Considerations: Unsaturated and Cyclic Carboxylic Acids
The principles discussed so far extend to more complex carboxylic acids.
Unsaturated Carboxylic Acids
Unsaturated carboxylic acids contain carbon-carbon double or triple bonds. The location of the double or triple bond is indicated using a number. The suffix "-enoic acid" is used for double bonds, and "-ynoic acid" is used for triple bonds.
Example: CH₂=CHCOOH is propenoic acid.
Cyclic Carboxylic Acids
Cyclic carboxylic acids have the carboxyl group attached to a ring. The ring is numbered, starting with the carboxyl carbon.
Example: A cyclopentane ring with a carboxylic acid group attached at carbon 1 is named cyclopentanecarboxylic acid.
Conclusion: Mastering IUPAC Nomenclature
Mastering IUPAC nomenclature for carboxylic acids is essential for success in organic chemistry. By following the systematic steps outlined in this guide and practicing with numerous examples, you'll confidently name even the most complex carboxylic acids. Remember to focus on identifying the parent chain, numbering correctly, and alphabetizing substituents properly. Consistent practice will refine your skills and build your understanding of this critical area of organic chemistry. With dedicated effort, naming carboxylic acids will transition from a challenging task to a routine and readily accomplished skill.
Latest Posts
Latest Posts
-
What Is The Temperature Of A Substance A Measure Of
Apr 04, 2025
-
Which Of The Following Are True About Algae
Apr 04, 2025
-
Orange Juice With Pulp Is A Heterogeneous Mixture
Apr 04, 2025
-
What Is The Most Widespread Tissue In The Body
Apr 04, 2025
-
What Is The Distance Between Saturn And The Sun
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Give The Iupac Name Of The Carboxylic Acid Below. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
