What Is The Charge On A Sulfide Ion
News Leon
Apr 01, 2025 · 5 min read
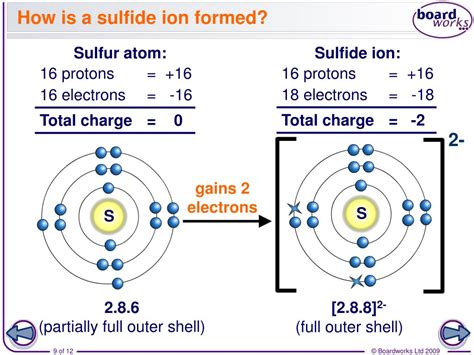
Table of Contents
What is the Charge on a Sulfide Ion? A Deep Dive into Sulfur's Anionic Behavior
The question, "What is the charge on a sulfide ion?" seems deceptively simple. However, understanding the answer requires a journey into the fascinating world of chemical bonding, electron configuration, and the periodic properties of elements. This comprehensive guide will explore the charge of the sulfide ion, its formation, and its significance in various chemical contexts. We'll delve into the underlying principles, explaining the concept in a way that's both accessible and insightful, enriching your understanding of basic chemistry.
Understanding Ions and Ionic Charge
Before diving into the specifics of the sulfide ion, let's establish a foundational understanding of ions and ionic charges. An ion is an atom or molecule that carries an electric charge. This charge arises from an imbalance in the number of protons (positively charged particles) and electrons (negatively charged particles).
-
Cations: Ions with a positive charge are called cations. They are formed when an atom loses one or more electrons. Metals typically form cations.
-
Anions: Ions with a negative charge are called anions. They are formed when an atom gains one or more electrons. Nonmetals typically form anions.
The magnitude of the ionic charge is represented by a numerical value with a plus or minus sign. For instance, a +2 charge indicates that the ion has lost two electrons, while a -1 charge indicates that it has gained one electron.
Sulfur's Position in the Periodic Table and its Electron Configuration
Sulfur (S), a nonmetal located in Group 16 (also known as the chalcogens) of the periodic table, plays a crucial role in understanding the sulfide ion's charge. Its atomic number is 16, meaning it has 16 protons and, in its neutral state, 16 electrons. The electron configuration of sulfur is 1s²2s²2p⁶3s²3p⁴. This configuration implies that the outermost shell (the valence shell) contains six electrons.
The Formation of the Sulfide Ion (S²⁻)
Sulfur's quest for stability drives its reaction to gain electrons. Atoms are most stable when their outermost electron shell is completely filled. For sulfur, achieving a stable octet (eight electrons in its valence shell) requires gaining two electrons. This electron gain results in the formation of the sulfide ion (S²⁻).
The process can be represented as:
S + 2e⁻ → S²⁻
This equation shows that a neutral sulfur atom (S) gains two electrons (2e⁻) to become a sulfide ion (S²⁻) with a -2 charge. This negative charge signifies the excess of two electrons compared to the number of protons.
The Significance of the -2 Charge in Chemical Reactions
The -2 charge of the sulfide ion dictates its behavior and reactivity in chemical reactions. Because it carries a negative charge, it is strongly attracted to positively charged ions (cations). This electrostatic attraction is the foundation of ionic bonding, a primary type of chemical bond.
Sulfide ions readily form ionic compounds with various metals. These compounds, known as sulfides, are numerous and exhibit diverse properties. For example:
- Iron(II) sulfide (FeS): A dark, crystalline solid commonly found in nature as the mineral pyrite.
- Sodium sulfide (Na₂S): A water-soluble compound used in various industrial processes.
- Lead(II) sulfide (PbS): A dark, insoluble compound, the principal component of the mineral galena.
The -2 charge of the sulfide ion significantly influences the properties of these sulfide compounds, including their solubility, melting points, and electrical conductivity.
Sulfide in Different Chemical Environments
The sulfide ion's behavior can be influenced by the chemical environment. In aqueous solutions, the sulfide ion can react with water molecules, undergoing hydrolysis to form hydrosulfide ions (HS⁻) and hydroxide ions (OH⁻). The extent of this hydrolysis depends on the pH of the solution.
Furthermore, the sulfide ion can act as a ligand in coordination complexes. A ligand is an ion or molecule that bonds to a central metal ion. Sulfide's ability to donate electron pairs makes it a versatile ligand capable of forming stable complexes with various transition metals. These complexes play vital roles in biological systems and industrial catalysis.
Applications of Sulfide Compounds
The unique properties of sulfide ions and their resulting compounds lead to a wide range of applications:
-
Mining and Metallurgy: Sulfide minerals are important sources of many metals. Extracting these metals often involves processes that exploit the chemical properties of the sulfide ions.
-
Industrial Chemistry: Sulfides are used in various industrial processes, including the production of chemicals, pigments, and fertilizers.
-
Environmental Science: Understanding the behavior of sulfide ions is crucial in environmental remediation efforts, particularly in addressing acid mine drainage, where sulfide minerals react with oxygen and water to produce sulfuric acid.
-
Biology: Sulfide ions play a role in certain biological processes, particularly in anaerobic environments. For instance, some microorganisms use sulfates (SO₄²⁻) as an electron acceptor during respiration, reducing them to sulfides.
Distinguishing Sulfide from Other Sulfur Anions
It's crucial to distinguish the sulfide ion (S²⁻) from other sulfur-containing anions, such as:
- Sulfite (SO₃²⁻): Contains sulfur bonded to three oxygen atoms, carrying a -2 charge.
- Sulfate (SO₄²⁻): Contains sulfur bonded to four oxygen atoms, also carrying a -2 charge.
- Thiosulfate (S₂O₃²⁻): Contains two sulfur atoms and three oxygen atoms, with a -2 charge.
Each of these anions exhibits distinct chemical properties due to differences in their structure and bonding. Their reactivity and applications differ substantially, highlighting the importance of correctly identifying the specific sulfur-containing anion involved.
Advanced Considerations: Beyond the Simple -2 Charge
While the -2 charge is the dominant oxidation state for sulfur in sulfide compounds, there are instances where sulfur's behavior is more complex. Under specific reaction conditions, sulfur can exhibit other oxidation states, including -1, 0, +2, +4, and +6. These alternative states often involve bonding with different atoms or the formation of polyatomic sulfur species. Understanding these complexities necessitates a deeper exploration into the principles of redox chemistry and advanced inorganic chemistry.
Conclusion: The Ubiquitous Sulfide Ion
The sulfide ion, with its characteristic -2 charge, plays a fundamental role in various aspects of chemistry. From the formation of ionic compounds to its significance in biological systems and industrial processes, understanding its behavior is crucial for appreciating the rich diversity of chemical interactions. This comprehensive exploration has provided a solid foundation for grasping the charge of the sulfide ion and its broader chemical implications, paving the way for further explorations into more advanced chemical concepts. Remember, chemistry is a journey of discovery, and the sulfide ion is just one intriguing piece of the puzzle.
Latest Posts
Latest Posts
-
Why Is Bone Considered A Connective Tissue
Apr 03, 2025
-
Why Is Dna Replication Considered Semi Conservative
Apr 03, 2025
-
Draw A Line Segment Of 7 2 Cm And Bisect It
Apr 03, 2025
-
In Parallelogram Abcd What Is Dc
Apr 03, 2025
-
What Part Of The Radius Articulates With The Humerus
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Charge On A Sulfide Ion . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
