What Happens To Plant Cells In A Hypertonic Solution
News Leon
Apr 05, 2025 · 5 min read
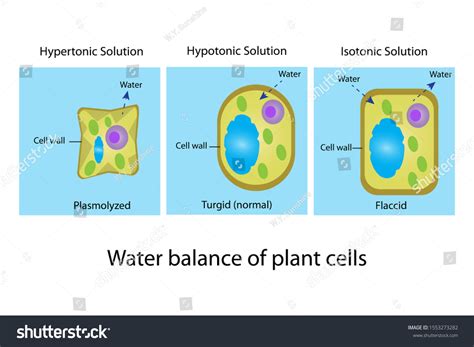
Table of Contents
What Happens to Plant Cells in a Hypertonic Solution? A Deep Dive into Plasmolysis
Plant cells, unlike animal cells, possess a rigid cell wall surrounding the cell membrane. This structural difference significantly impacts how they respond to different osmotic environments, particularly hypertonic solutions. Understanding this response is crucial in various fields, from agriculture and horticulture to plant physiology research. This article will comprehensively explore the process of plasmolysis, detailing the changes plant cells undergo in a hypertonic solution and the underlying mechanisms involved.
Understanding Osmosis and Tonicity
Before diving into the specifics of plant cell behavior, let's establish a foundation in osmosis and tonicity. Osmosis is the passive movement of water across a selectively permeable membrane from a region of high water potential (low solute concentration) to a region of low water potential (high solute concentration). This movement continues until equilibrium is reached, or the water potential is equal on both sides of the membrane.
Tonicity describes the relative concentration of solutes in two solutions separated by a selectively permeable membrane. There are three main types of tonicity:
- Hypotonic: The solution outside the cell has a lower solute concentration (and therefore a higher water potential) than the solution inside the cell.
- Isotonic: The solution outside the cell has the same solute concentration as the solution inside the cell.
- Hypertonic: The solution outside the cell has a higher solute concentration (and therefore a lower water potential) than the solution inside the cell. This is the focus of our exploration.
Plasmolysis: The Effect of a Hypertonic Solution on Plant Cells
When a plant cell is placed in a hypertonic solution, water moves out of the cell via osmosis. This is because the water potential inside the cell is higher than the water potential outside the cell. The water leaves the cell's central vacuole, which is responsible for maintaining turgor pressure – the pressure exerted by the cell contents against the cell wall. As water exits, the vacuole shrinks, and the cell membrane pulls away from the cell wall. This process is known as plasmolysis.
Stages of Plasmolysis
Plasmolysis doesn't happen instantaneously. It's a gradual process that can be observed in several stages:
- Incipient Plasmolysis: This is the initial stage where the plasma membrane begins to pull away from the cell wall at the corners or edges of the cell. It's often subtle and might require microscopy to detect. At this point, the cell is still relatively turgid.
- Plasmolysis: As water continues to exit, the plasma membrane pulls away further from the cell wall, creating a noticeable gap between the two. This gap is filled with the hypertonic solution. The cell loses its turgidity and appears flaccid.
- Complete Plasmolysis: In this advanced stage, the plasma membrane is completely detached from the cell wall. The cell is significantly shrunken and appears plasmolyzed. The cell's shape is distorted, and its metabolic activity is often impaired.
Factors Influencing Plasmolysis
The rate and extent of plasmolysis are influenced by several factors:
- Concentration of the Hypertonic Solution: A higher concentration of solute in the external solution will lead to a faster and more pronounced plasmolysis.
- Type of Solute: The nature of the solute can also affect plasmolysis. Some solutes may penetrate the cell membrane, altering the osmotic gradient and affecting the water movement.
- Cell Wall Properties: The rigidity of the cell wall influences the extent of plasmolysis. A more rigid cell wall will offer some resistance to the shrinking of the cell.
- Temperature: Temperature affects the rate of osmosis; higher temperatures generally lead to faster water movement.
- Plant Species: Different plant species may exhibit varying degrees of sensitivity to hypertonic solutions, reflecting differences in their cell wall structure and membrane permeability.
The Role of the Cell Wall in Plasmolysis
The presence of the cell wall is what distinguishes plasmolysis in plant cells from the similar process of crenation in animal cells. The cell wall provides structural support and prevents the cell from completely collapsing even during significant water loss. However, the cell wall cannot indefinitely resist the osmotic pressure. If the cell remains in the hypertonic solution for an extended period, irreversible damage can occur.
Reversal of Plasmolysis: Deplasmolysis
The process of plasmolysis is reversible if the plant cell is transferred to a hypotonic or isotonic solution. Water will then move back into the cell via osmosis, causing the cell to regain its turgor pressure and the plasma membrane to adhere to the cell wall again. This process is called deplasmolysis.
Consequences of Plasmolysis
While plasmolysis is a reversible process under certain conditions, prolonged exposure to a hypertonic solution can lead to several detrimental consequences for plant cells:
- Reduced Turgor Pressure: The loss of turgor pressure affects the plant's overall structure and can lead to wilting. This is often observed in plants experiencing drought stress.
- Impaired Metabolic Processes: The disruption of cellular organization and decreased water availability can negatively impact various metabolic processes, including photosynthesis and respiration.
- Cell Death: In severe cases of plasmolysis, irreversible damage can occur, leading to the death of the plant cell. The prolonged shrinkage and dehydration can severely disrupt cellular integrity and functionality.
Practical Applications and Significance
Understanding plasmolysis is essential in several areas:
- Agriculture and Horticulture: Knowing how plants respond to hypertonic conditions is vital for managing irrigation, soil salinity, and fertilizer application. Effective water management is crucial for optimal plant growth and yield.
- Plant Physiology Research: Studying plasmolysis helps researchers understand plant responses to environmental stress and develop strategies for improving crop tolerance to drought and salinity.
- Food Preservation: Plasmolysis is used in food preservation techniques, like pickling and jam-making, where high solute concentrations prevent microbial growth by inhibiting their metabolism.
- Medicine: Some studies explore the application of hypertonic solutions in treating certain medical conditions, and understanding their effects on cells is crucial.
Conclusion
Plasmolysis is a fascinating process that demonstrates the intricate relationship between plant cells and their environment. The ability to withstand or recover from hypertonic conditions is a key aspect of plant survival and adaptation. By understanding the mechanisms of plasmolysis and its consequences, we can develop better strategies for managing plant growth and addressing challenges posed by environmental stress. Further research in this area promises to yield valuable insights for improving agricultural practices and enhancing our understanding of plant biology. Future research may focus on the role of specific genes and proteins in the plasmolysis process and in determining the degree of tolerance among different plant species. The continuing exploration of plasmolysis will undoubtedly contribute to advancements in numerous fields related to plant science and beyond.
Latest Posts
Latest Posts
-
Its Condyles Articulate With The Atlas
Apr 05, 2025
-
Which Of The Following Equations Are Identities
Apr 05, 2025
-
Sugar Dissolved In Water Physical Or Chemical Change
Apr 05, 2025
-
12 Is 75 Of What Number
Apr 05, 2025
-
Ammonium Chloride Formula By Criss Cross Method
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Happens To Plant Cells In A Hypertonic Solution . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
