Ammonium Chloride Formula By Criss Cross Method
News Leon
Apr 05, 2025 · 6 min read
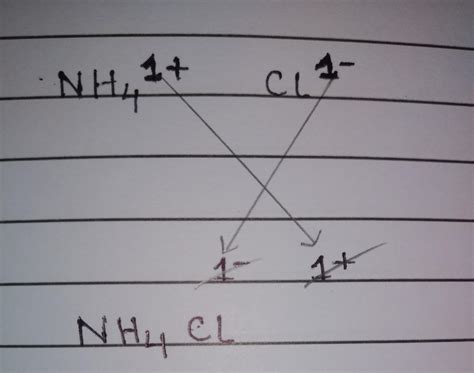
Table of Contents
Ammonium Chloride Formula: A Deep Dive into the Criss-Cross Method
Ammonium chloride, a fascinating inorganic compound with a wide array of applications, provides an excellent example of how the criss-cross method simplifies determining chemical formulas. This method, particularly useful for ionic compounds, allows us to predict the formula based on the charges of the constituent ions. This comprehensive guide will delve into the intricacies of ammonium chloride's formula derivation using the criss-cross method, exploring its properties, applications, and related concepts.
Understanding the Criss-Cross Method
The criss-cross method is a straightforward technique used to predict the chemical formula of an ionic compound formed by a cation (positively charged ion) and an anion (negatively charged ion). The absolute values of the charges are exchanged and become the subscripts of the respective ions in the chemical formula. Let's break this down step-by-step:
-
Identify the Ions: First, you need to identify the cation and the anion involved in the compound. For ammonium chloride, we have the ammonium ion (NH₄⁺) and the chloride ion (Cl⁻).
-
Determine the Charges: Next, determine the charge of each ion. Ammonium (NH₄⁺) carries a +1 charge, while chloride (Cl⁻) carries a -1 charge.
-
Criss-Cross the Charges: This is where the "criss-cross" comes in. The absolute value of the charge of each ion becomes the subscript of the other ion. So, the +1 charge of ammonium becomes the subscript of chloride, and the -1 charge of chloride becomes the subscript of ammonium.
-
Simplify the Formula (if necessary): After criss-crossing, simplify the subscripts to the smallest whole number ratio. In the case of ammonium chloride, we have (NH₄)₁Cl₁, which simplifies to NH₄Cl.
Applying the Criss-Cross Method to Ammonium Chloride
Let's illustrate the criss-cross method for ammonium chloride specifically:
Step 1: Identify the ions:
- Cation: Ammonium ion (NH₄⁺)
- Anion: Chloride ion (Cl⁻)
Step 2: Determine the charges:
- Ammonium ion charge: +1
- Chloride ion charge: -1
Step 3: Criss-cross the charges:
- The +1 charge of NH₄⁺ becomes the subscript of Cl⁻.
- The -1 charge of Cl⁻ becomes the subscript of NH₄⁺.
This gives us the preliminary formula: (NH₄)₁Cl₁
Step 4: Simplify the formula: Since the subscripts are already in the simplest whole-number ratio (1:1), we don't need to simplify further.
Therefore, the final formula for ammonium chloride is NH₄Cl.
Properties of Ammonium Chloride
Ammonium chloride (NH₄Cl) is a white crystalline salt that is highly soluble in water. It exhibits several key properties:
- Solubility: Readily dissolves in water, exhibiting endothermic dissolution (absorbs heat).
- Acidity: It's an acidic salt, meaning its aqueous solution is acidic due to the hydrolysis of the ammonium ion.
- Crystal Structure: Possesses a cubic crystal structure.
- Sublimation: It can sublime, meaning it can transition directly from the solid phase to the gaseous phase without passing through the liquid phase. This occurs at relatively low temperatures.
- Thermal Decomposition: At higher temperatures, it decomposes to produce ammonia gas (NH₃) and hydrogen chloride gas (HCl).
Applications of Ammonium Chloride
Ammonium chloride finds a wide range of applications across various industries, including:
- Fertilizers: A major application is as a nitrogen fertilizer, providing a crucial nutrient for plant growth.
- Medicine: Used as an expectorant in cough medicines to help loosen phlegm. It's also used in some electrolyte solutions.
- Food Industry: Used as a food additive in some instances, acting as a yeast nutrient and an acidity regulator.
- Electroplating: Acts as an electrolyte in some electroplating processes.
- Metallurgy: Used as a flux in metal welding and soldering, aiding in the cleaning of metal surfaces.
- Batteries: A component in some types of batteries.
- Dry Cell Batteries: Ammonium chloride is an important part of the electrolyte in dry-cell batteries, contributing to their functionality. The reaction between ammonium chloride and zinc creates an electron flow that produces electricity.
Safety Precautions and Handling
While generally safe when handled appropriately, ammonium chloride presents certain safety concerns:
- Inhalation: Inhalation of ammonium chloride dust can cause respiratory irritation. Appropriate respiratory protection should be used in handling powdered ammonium chloride.
- Skin and Eye Contact: Contact with skin or eyes can cause irritation. Immediate flushing with water is necessary in case of contact.
- Ingestion: Ingestion of ammonium chloride can cause gastrointestinal distress.
Beyond the Criss-Cross Method: Understanding Ionic Bonding
The criss-cross method is a valuable shortcut, but it's crucial to understand the underlying principles of ionic bonding. Ammonium chloride's formation is a consequence of the electrostatic attraction between the positively charged ammonium ion (NH₄⁺) and the negatively charged chloride ion (Cl⁻).
The ammonium ion itself is a polyatomic ion, formed from a covalent bond between one nitrogen atom and four hydrogen atoms. The nitrogen atom shares electrons with each hydrogen atom to achieve a stable octet. The entire ammonium group then carries a positive charge because the nitrogen atom has effectively lost one electron.
The chloride ion is formed when a chlorine atom gains one electron, achieving a stable octet and acquiring a negative charge.
The strong electrostatic attraction between the positively charged ammonium ion and the negatively charged chloride ion results in the formation of the ionic compound ammonium chloride. This attraction forms a crystalline lattice structure, holding the ions together in a repeating pattern.
Related Concepts and Further Exploration
Several related concepts provide further context for understanding ammonium chloride and the criss-cross method:
- Polyatomic Ions: The ammonium ion (NH₄⁺) is a polyatomic ion, highlighting the fact that the criss-cross method applies equally to compounds containing single atoms and polyatomic ions.
- Electrolytes: Ammonium chloride's ability to conduct electricity when dissolved in water underscores its role as an electrolyte. The dissolved ions carry the electric current.
- Acid-Base Reactions: Ammonium chloride’s acidic nature highlights its participation in acid-base reactions. Its acidic behavior is due to the ammonium ion’s tendency to donate a proton (H⁺).
- Solubility Rules: Understanding solubility rules can help predict whether a given ionic compound will dissolve in water.
Conclusion
The criss-cross method provides a simple and effective way to determine the chemical formula of ionic compounds, such as ammonium chloride. This approach, however, should be complemented by a deeper understanding of ionic bonding, polyatomic ions, and the underlying chemical principles governing compound formation. Ammonium chloride, with its versatile applications and interesting properties, serves as a prime example to solidify this understanding. The safety precautions associated with its handling should also be consistently followed to ensure safe usage and prevent any harmful effects. Further exploration of the related concepts mentioned above will enhance the comprehensive understanding of this crucial inorganic compound.
Latest Posts
Latest Posts
-
What Is At The Base Of All Ecological Pyramids
Apr 05, 2025
-
A Bar Magnet Is Placed In A Uniform Magnetic Field
Apr 05, 2025
-
Balance Equation Naoh H2so4 Na2so4 H2o
Apr 05, 2025
-
Non Metal Liquid At Room Temp
Apr 05, 2025
-
Which Of The Following Is An Example Of Pollution
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Ammonium Chloride Formula By Criss Cross Method . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
