What Happens To A Liquid When Heated
News Leon
Apr 05, 2025 · 5 min read
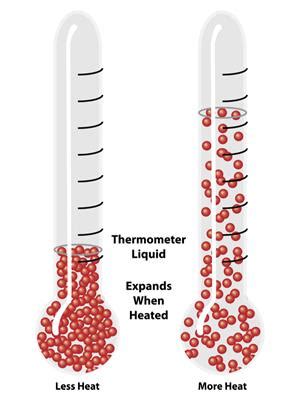
Table of Contents
What Happens to a Liquid When Heated? A Deep Dive into Thermal Properties
Heating a liquid might seem like a simple process, but the underlying physics and chemistry are surprisingly complex and fascinating. Understanding what happens to a liquid when heated is crucial in numerous fields, from cooking and material science to meteorology and industrial processes. This comprehensive article will delve into the various changes a liquid undergoes upon heating, exploring the concepts of temperature, heat transfer, phase transitions, and the influence of intermolecular forces.
The Fundamentals: Temperature and Heat
Before we explore the effects of heating on liquids, let's clarify the difference between temperature and heat. Temperature is a measure of the average kinetic energy of the particles (atoms or molecules) within a substance. Higher temperature means the particles are moving faster and possess more kinetic energy. Heat, on the other hand, is the transfer of energy from a hotter object to a colder object. When you heat a liquid, you're transferring thermal energy to its particles, increasing their average kinetic energy and thus raising its temperature.
Heat Transfer Mechanisms
The transfer of heat to a liquid can occur through three primary mechanisms:
- Conduction: Heat transfer through direct contact. The heated container transfers thermal energy directly to the liquid molecules in contact with its surface.
- Convection: Heat transfer through the movement of fluids. As the liquid near the heat source warms up, it becomes less dense and rises, while cooler, denser liquid sinks to take its place, creating a cycle of heat transfer.
- Radiation: Heat transfer through electromagnetic waves. While less significant in heating liquids directly, radiation from a heat source (like a burner) can contribute to the overall heating process.
Expansion and Density Changes
One of the most immediate and observable effects of heating a liquid is its expansion. As the kinetic energy of the liquid's particles increases, they move more vigorously, leading to an increase in the average distance between them. This results in an overall increase in the liquid's volume. Conversely, the density of the liquid decreases because the same mass now occupies a larger volume. The degree of expansion varies depending on the specific liquid and the temperature range.
Thermal Expansion Coefficient
The thermal expansion coefficient is a property that quantifies the extent to which a material expands or contracts with a change in temperature. Different liquids have different thermal expansion coefficients. Water, for instance, exhibits an anomalous behavior near its freezing point, contracting upon heating until it reaches 4°C before expanding. This unusual property has significant implications for aquatic life and the Earth's climate.
Changes in Viscosity and Surface Tension
Heating a liquid also affects its viscosity, which is a measure of its resistance to flow. Higher temperatures generally lead to lower viscosity because the increased kinetic energy of the molecules reduces the intermolecular forces that hinder their movement. This is why, for example, honey flows more easily when warm than when cold.
Similarly, heating a liquid often reduces its surface tension. Surface tension is the tendency of liquid surfaces to minimize their area, creating a "skin-like" effect. Higher temperatures weaken the intermolecular forces responsible for surface tension, causing the surface to become less taut.
Phase Transitions: Boiling and Vaporization
Perhaps the most dramatic change that occurs when a liquid is heated is its phase transition. As the temperature continues to rise, the liquid eventually reaches its boiling point. At this point, the kinetic energy of the molecules is sufficient to overcome the intermolecular forces holding them together in the liquid phase. The liquid begins to boil, transforming into a gas (vapor) through a process called vaporization.
Boiling Point and Pressure
The boiling point of a liquid is not a fixed value; it depends on the pressure exerted on the liquid. At higher pressures, the boiling point increases, as the molecules require more energy to overcome the greater external forces. This is why a pressure cooker can cook food faster; the increased pressure raises the boiling point of water, allowing it to reach higher temperatures. Conversely, at lower pressures, the boiling point decreases.
Evaporation vs. Boiling
It's important to differentiate between boiling and evaporation. Boiling is a bulk process occurring throughout the liquid at its boiling point, while evaporation is a surface phenomenon that can occur at any temperature below the boiling point. Evaporation is driven by the escape of high-energy molecules from the liquid's surface. The rate of evaporation increases with temperature, surface area, and air movement.
Chemical Changes: Decomposition and Reactions
While heating a liquid primarily leads to physical changes, it can also induce chemical changes under certain conditions. Some liquids may decompose at high temperatures, breaking down into simpler substances. Others might undergo chemical reactions with dissolved substances or with the container material itself. The specific chemical changes depend heavily on the composition of the liquid and the heating conditions.
Examples of Chemical Changes
- Decomposition of Hydrogen Peroxide: Heating hydrogen peroxide can decompose it into water and oxygen gas.
- Thermal Degradation of Polymers: Many polymers undergo thermal degradation at high temperatures, breaking down their long-chain structures.
- Reactions in Solutions: Heating solutions can accelerate chemical reactions between dissolved substances.
Applications and Real-World Examples
Understanding the effects of heating on liquids is crucial in a wide range of applications:
- Cooking: Heating liquids is fundamental to cooking. Boiling water, simmering sauces, and frying food all involve the principles of heat transfer and phase transitions.
- Industrial Processes: Many industrial processes, such as distillation, evaporation, and sterilization, rely on heating liquids to achieve desired outcomes.
- Climate Science: Understanding the thermal properties of water is vital for understanding climate patterns, weather systems, and ocean currents.
- Material Science: The thermal expansion and viscosity of liquids play crucial roles in the manufacturing and processing of various materials.
Conclusion
Heating a liquid is a multifaceted process with far-reaching consequences. From the simple expansion and changes in viscosity to the dramatic phase transitions and potential chemical reactions, the effects of heat on liquids are diverse and fascinating. Understanding these effects is critical in numerous scientific and technological applications, making this a topic deserving of continued exploration and research. The interplay between temperature, heat transfer, intermolecular forces, and the properties of specific liquids creates a rich tapestry of physical and chemical phenomena that continue to intrigue scientists and engineers alike. Further research into the specific behavior of different liquids under varying heating conditions will continue to unlock new insights and opportunities for innovation.
Latest Posts
Latest Posts
-
In The Carbon Cycle The Role Of Plants Is To
Apr 06, 2025
-
What Do All Elements In A Group Have In Common
Apr 06, 2025
-
How Many Light Years Away Is Mars From Earth
Apr 06, 2025
-
Which Of The Following Is Not A Characteristic Of Enzymes
Apr 06, 2025
-
Why Are Maps Important To Geographers
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Happens To A Liquid When Heated . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
