What Do All Elements In A Group Have In Common
News Leon
Apr 06, 2025 · 6 min read
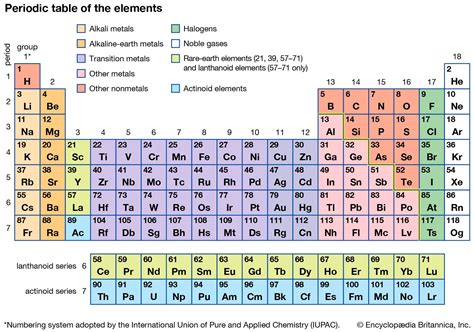
Table of Contents
What Do All Elements in a Group Have in Common? Understanding Periodic Table Trends
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding the relationships between elements within the same group, or column, is crucial for predicting their behavior and understanding their applications. This article delves deep into the commonalities of elements within each group, exploring their electronic configurations, chemical properties, and physical characteristics.
The Significance of Groupings: A Foundation for Understanding
The periodic table’s structure is not arbitrary. Elements are arranged in groups because they share fundamental similarities in their atomic structure, specifically their valence electrons. These are the electrons in the outermost shell, which are primarily responsible for an element's chemical reactivity. Elements in the same group have the same number of valence electrons, leading to similar chemical behaviors and bonding patterns. This shared characteristic forms the basis for understanding the trends and properties observed within each group.
Group 1: The Alkali Metals – Reactivity Personified
Alkali metals, including lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr), are all characterized by having one valence electron. This single electron is easily lost, making them highly reactive. Their reactivity increases as you go down the group, due to the increasing atomic size and decreasing ionization energy.
Key Characteristics of Alkali Metals:
- Highly reactive: They readily react with water, producing hydrogen gas and a metal hydroxide.
- Low density: They are soft, silvery-white metals with low densities.
- Low melting and boiling points: Compared to other metals, they have relatively low melting and boiling points.
- Form +1 ions: They readily lose their single valence electron to form ions with a +1 charge.
- Excellent conductors of electricity and heat: This is a consequence of their electronic structure.
Group 2: The Alkaline Earth Metals – Less Reactive, But Still Significant
The alkaline earth metals, comprising beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra), each possess two valence electrons. While less reactive than alkali metals, they are still reactive enough to participate in various chemical reactions. Their reactivity, like alkali metals, increases as you descend the group.
Key Characteristics of Alkaline Earth Metals:
- Reactive, but less than alkali metals: They react with water, but generally more slowly than alkali metals.
- Higher density and melting points: Compared to alkali metals, they have higher densities and melting points.
- Form +2 ions: They lose their two valence electrons to form ions with a +2 charge.
- Good conductors of electricity and heat: Similar to alkali metals, they are good conductors.
- Many important biological roles: For example, calcium is essential for bone formation.
Groups 13-18: Exploring the p-Block Elements
Groups 13 to 18 constitute the p-block elements, where the valence electrons occupy the p orbitals. Each group exhibits unique properties dictated by the number of valence electrons.
Group 13: The Boron Group – A Diverse Group
Boron (B), aluminum (Al), gallium (Ga), indium (In), and thallium (Tl) have three valence electrons. This group displays a wide range of properties, with boron being a metalloid and the rest being metals. Their chemistry is complex and influenced by the varying electronegativities of the elements.
Group 14: The Carbon Group – The Basis of Life
Carbon (C), silicon (Si), germanium (Ge), tin (Sn), and lead (Pb) possess four valence electrons. This group showcases the remarkable versatility of carbon in forming diverse organic compounds, as well as the semiconductor properties of silicon and germanium, crucial in electronics.
Group 15: The Nitrogen Group – Diverse Oxidation States
Nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), and bismuth (Bi) all have five valence electrons. This group displays a wide variety of oxidation states, reflecting the ability of these elements to gain, lose, or share electrons. Nitrogen, in particular, plays a vital role in biological systems.
Group 16: The Chalcogens – Essential Elements
Oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and polonium (Po) possess six valence electrons. This group includes oxygen, essential for respiration, and sulfur, crucial in various industrial processes. They tend to form -2 ions, except for oxygen which is almost always found as a diatomic molecule.
Group 17: The Halogens – Highly Reactive Nonmetals
Fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At) have seven valence electrons. This group consists of highly reactive nonmetals that readily gain an electron to form -1 ions, achieving a stable octet. Their reactivity decreases as you go down the group.
Group 18: The Noble Gases – Inert Elements
Helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn) have eight valence electrons (except helium with two), completing their outermost shell. This complete octet makes them exceptionally stable and unreactive, hence their designation as "noble gases."
Transition Metals: A Sea of Complexity
The transition metals occupy the central block of the periodic table. They are characterized by incompletely filled d orbitals and variable oxidation states. This characteristic leads to their diverse and often complex chemical behavior. Unlike the main group elements, predicting their properties solely based on their group number is less straightforward. Their properties are influenced by factors such as d-orbital interactions and ligand field effects. They show a wide range of colorful compounds and are often used as catalysts.
Inner Transition Metals: Lanthanides and Actinides
The lanthanides and actinides, located below the main body of the periodic table, are characterized by the filling of the f-orbitals. They exhibit similar chemical properties within their respective series, making it challenging to predict individual element behavior solely based on their group placement.
Trends Across Periods and Groups
Understanding the trends across both periods (rows) and groups is essential. Several key trends emerge:
- Atomic Radius: Atomic radius generally increases down a group as additional electron shells are added. It decreases across a period as the effective nuclear charge increases.
- Ionization Energy: Ionization energy, the energy required to remove an electron, generally decreases down a group as the outermost electrons are farther from the nucleus and less strongly held. It increases across a period due to the increasing effective nuclear charge.
- Electronegativity: Electronegativity, the ability of an atom to attract electrons in a chemical bond, generally decreases down a group and increases across a period.
- Metallic Character: Metallic character increases down a group (elements become more metallic) and decreases across a period (elements become less metallic).
Conclusion: A Unified Framework for Understanding Elements
The periodic table’s structure is a testament to the underlying order in the universe. The grouping of elements based on their shared number of valence electrons provides a powerful framework for understanding their chemical and physical properties. While nuances exist, particularly within the transition and inner transition metals, the fundamental principles of electron configuration and valence electrons remain central to predicting and understanding the behavior of elements in their respective groups. This understanding is crucial not only for fundamental chemical knowledge but also for the development of new materials and technologies. By recognizing the common threads that bind elements within each group, we gain a deeper appreciation for the interconnectedness of the chemical world.
Latest Posts
Latest Posts
-
Quotient Of A Number And 3
Apr 06, 2025
-
Write The Complete Nuclear Equation For The Bombardment
Apr 06, 2025
-
Geothermal Energy Is Derived From The Sun
Apr 06, 2025
-
Geometric Mean Of 9 And 16
Apr 06, 2025
-
Definition Of Initial Velocity In Physics
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Do All Elements In A Group Have In Common . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
