Value Representing The Number Of Protons In An Element
News Leon
Apr 04, 2025 · 7 min read
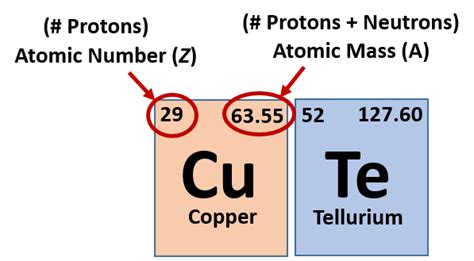
Table of Contents
Atomic Number: The Defining Characteristic of an Element
The atomic number, a seemingly simple whole number, holds the key to understanding the fundamental nature of elements. This critical value represents the number of protons found in the nucleus of an atom, defining its identity and its place on the periodic table. More than just a label, the atomic number dictates an element's chemical properties, its reactivity, and its behavior in various chemical reactions. Understanding the atomic number is fundamental to grasping the entire field of chemistry and its related sciences.
What is an Atomic Number?
The atomic number, often represented by the symbol Z, is a unique identifier for each chemical element. It represents the number of positively charged protons present within the atom's nucleus. This is crucial because the number of protons defines the element. No two elements have the same atomic number. For example, hydrogen (H) has an atomic number of 1 (one proton), helium (He) has an atomic number of 2 (two protons), lithium (Li) has an atomic number of 3 (three protons), and so on. This sequential increase in the number of protons forms the basis of the periodic table's organization.
It's important to differentiate the atomic number from other atomic properties. While the number of neutrons (neutral particles in the nucleus) can vary within an element (creating isotopes), and the number of electrons (negatively charged particles orbiting the nucleus) can change (forming ions), the number of protons remains constant and defines the element's identity. Changing the number of protons fundamentally changes the element itself.
The Significance of Protons
Protons, along with neutrons, constitute the atom's nucleus, accounting for the vast majority of its mass. However, their significance extends far beyond their mass contribution. The positive charge of the protons determines the overall positive charge of the nucleus. This positive charge is what attracts the negatively charged electrons, holding them in orbit and forming the atom's structure.
The arrangement of electrons in energy levels and orbitals, directly influenced by the number of protons, dictates the element's chemical behavior. Electrons in the outermost shell, called valence electrons, are primarily responsible for an element's reactivity. The number of valence electrons is directly related to the atomic number and, therefore, the element's position in the periodic table. This connection allows us to predict an element's reactivity based solely on its atomic number.
The Periodic Table and Atomic Number
The periodic table is organized in a way that directly reflects the increasing atomic number of elements. Elements are arranged sequentially from left to right and top to bottom based on their Z value. This arrangement is not arbitrary; it results in elements with similar chemical properties falling into the same groups (columns) and periods (rows). This organization is one of the most significant achievements in the history of chemistry and allows us to predict the behavior of elements based on their position on the table.
Groups and Periods: Reflecting Electron Configurations
Elements in the same group share similar chemical properties because they have the same number of valence electrons. These valence electrons are the primary players in chemical bonding, determining how an element will interact with other elements. The atomic number provides the foundation for understanding the electron configuration, which, in turn, explains the chemical behavior.
Periods represent the successive filling of electron shells. As you move across a period, the atomic number increases, adding one proton and, usually, one electron. This electron fills the existing energy levels before starting a new shell in the next period. This regular filling pattern accounts for the periodic trends in properties observed in the periodic table, such as electronegativity, ionization energy, and atomic radius.
Isotopes and Atomic Number
While the atomic number remains constant for a given element, the number of neutrons can vary, leading to the existence of isotopes. Isotopes are atoms of the same element (same atomic number) but with different numbers of neutrons. This means they have the same number of protons but a different mass number (A), which is the sum of protons and neutrons.
For example, carbon (C) has an atomic number of 6. The most common isotope is carbon-12 (¹²C), with 6 protons and 6 neutrons. However, carbon-13 (¹³C) and carbon-14 (¹⁴C) also exist, each with 6 protons but 7 and 8 neutrons, respectively. Despite having different numbers of neutrons, all these are still carbon atoms because their atomic number (number of protons) remains 6. The difference in neutron number affects the mass and sometimes the stability (radioactivity) of the isotope, but not the fundamental chemical properties determined by the atomic number.
Impact of Isotopes on Atomic Mass
The variation in the number of neutrons among isotopes leads to a slightly different average atomic mass for each element. The atomic mass listed on the periodic table is a weighted average of the masses of all naturally occurring isotopes of an element, accounting for their relative abundance. This average mass is crucial for various chemical calculations and is not directly related to the atomic number, which remains constant regardless of isotopic variation.
Atomic Number and Chemical Properties
The atomic number is the single most important factor determining an element's chemical properties. These properties describe how an element behaves in chemical reactions, including its ability to form bonds, its reactivity, and the types of compounds it can form.
Chemical Bonding and Reactivity
The number of valence electrons, directly related to the atomic number, determines an element's reactivity. Elements with a nearly full or empty valence shell are generally less reactive (e.g., noble gases), while those with partially filled valence shells tend to be more reactive (e.g., alkali metals and halogens). The atomic number allows us to predict the number of bonds an atom can form, the type of bonds (ionic, covalent, metallic), and the overall chemical behavior.
Oxidation States and Chemical Reactions
The atomic number also influences an element's oxidation state, representing the charge an atom has or appears to have in a compound. This oxidation state is crucial in predicting the products and stoichiometry of chemical reactions. For example, understanding the atomic number of elements helps predict whether a reaction will be redox (oxidation-reduction) and the potential transfer of electrons between elements.
Atomic Number and Nuclear Chemistry
The atomic number plays a critical role in understanding nuclear reactions. Nuclear reactions involve changes in the nucleus of an atom, resulting in a change in the number of protons or neutrons, or both. These changes can transform one element into another.
Radioactive Decay and Atomic Number
Radioactive decay is a process where unstable atomic nuclei spontaneously emit particles or energy. Certain types of radioactive decay alter the atomic number of the nucleus, effectively transforming the element. For example, alpha decay reduces the atomic number by 2, while beta decay increases the atomic number by 1. Understanding the atomic number is essential for analyzing and predicting the products of radioactive decay.
Nuclear Fission and Fusion
Nuclear fission involves splitting a heavy nucleus into smaller nuclei, and nuclear fusion involves combining light nuclei to form a heavier nucleus. Both processes involve changes in the atomic number and release vast amounts of energy. Understanding the atomic number is critical in understanding the stability of nuclei and the feasibility of nuclear reactions.
Applications of Atomic Number Knowledge
The knowledge of atomic numbers has far-reaching applications across numerous fields, including:
- Chemistry: Understanding chemical reactions, predicting reactivity, and determining the properties of compounds.
- Physics: Analyzing nuclear reactions, studying atomic structure, and developing new technologies based on atomic properties.
- Material Science: Designing new materials with specific properties based on the properties of elements and their combinations.
- Medicine: Using radioactive isotopes for medical imaging and treatment (e.g., PET scans, radiotherapy).
- Environmental Science: Tracing pollutants and understanding environmental processes using isotopic analysis.
- Archaeology: Dating ancient artifacts using radiocarbon dating, which relies on the decay of carbon-14.
Conclusion: The Foundational Role of Atomic Number
The atomic number, representing the number of protons in an atom's nucleus, is more than just a simple number; it's a fundamental property that dictates an element's identity, chemical behavior, and role in various processes. Its significance spans across multiple scientific disciplines, making it a cornerstone concept for understanding the physical world. From the organization of the periodic table to the intricacies of nuclear reactions, the atomic number provides a unifying framework for understanding the nature of matter and its interactions. Its importance continues to drive advancements in numerous fields, highlighting its enduring relevance in science and technology. A deep understanding of atomic number is therefore indispensable for anyone seeking to explore the fascinating world of chemistry and beyond.
Latest Posts
Latest Posts
-
Most Widely Distributed Tissue Type In The Body
Apr 04, 2025
-
Which Is The Correct Electron Configuration For Arsenic
Apr 04, 2025
-
Oils Are Liquid At Room Temperature Because
Apr 04, 2025
-
9 To The Power Of 3 2
Apr 04, 2025
-
Volume Of 1 Drop Of Water
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Value Representing The Number Of Protons In An Element . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
