Which Is The Correct Electron Configuration For Arsenic
News Leon
Apr 04, 2025 · 6 min read
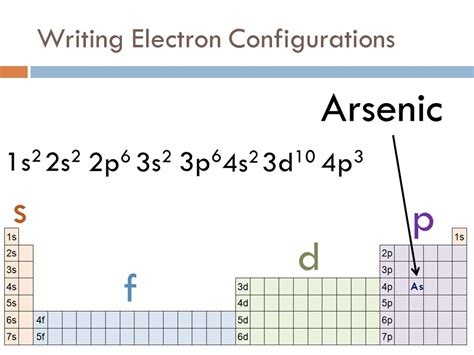
Table of Contents
Which is the Correct Electron Configuration for Arsenic? Understanding Electron Shell Filling and Exceptions
Arsenic, a metalloid with the atomic number 33, presents a fascinating case study in electron configuration, highlighting the complexities and exceptions to the standard Aufbau principle. While a simplistic approach might suggest a straightforward electron arrangement, a deeper understanding reveals nuances crucial for accurate representation and predicting arsenic's chemical behavior. This article delves into the intricacies of arsenic's electron configuration, exploring the underlying principles, explaining potential discrepancies, and ultimately establishing the correct and most widely accepted representation.
Understanding Electron Configuration Basics
Before diving into the specifics of arsenic, let's briefly revisit the fundamental principles governing electron configuration. The electron configuration describes the arrangement of electrons within an atom's energy levels and sublevels. These energy levels are denoted by principal quantum numbers (n = 1, 2, 3, etc.), with each level containing sublevels (s, p, d, and f). Each sublevel can hold a specific number of electrons:
- s sublevel: Holds a maximum of 2 electrons.
- p sublevel: Holds a maximum of 6 electrons.
- d sublevel: Holds a maximum of 10 electrons.
- f sublevel: Holds a maximum of 14 electrons.
The Aufbau principle dictates that electrons fill the lowest energy levels first, proceeding to higher energy levels only after lower ones are completely filled. The Hund's rule states that electrons will individually occupy each orbital within a subshell before doubling up in any one orbital. Finally, the Pauli exclusion principle asserts that no two electrons within an atom can have the same four quantum numbers (n, l, ml, and ms).
The Standard Approach and its Limitations: Arsenic's Expected Configuration
Based on the Aufbau principle, one might expect arsenic (atomic number 33) to have the following electron configuration:
1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p³
This configuration follows the order of filling: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p. It accounts for all 33 electrons of arsenic. However, this seemingly straightforward configuration doesn't fully capture the complexities of electron-electron interactions within the atom.
Orbital Energies and the Subtleties of Electron Interactions
The simple Aufbau principle assumes that the energy levels of orbitals are strictly ordered. However, this is not entirely accurate. The energies of orbitals can be influenced by factors such as electron shielding and interelectronic repulsion. These effects can cause orbital energies to deviate from the expected order, leading to exceptions in electron configurations. Specifically, the energy difference between the 4s and 3d orbitals is relatively small. In some cases, filling the 3d orbital before the 4s can result in a more stable configuration.
The Actual (and Correct) Electron Configuration for Arsenic
While the simplified Aufbau configuration is a good starting point, the actual electron configuration of arsenic is slightly different. It's more accurately represented as:
[Ar] 3d¹⁰ 4s² 4p³
Where [Ar] represents the electron configuration of Argon (1s²2s²2p⁶3s²3p⁶), which is a noble gas with a stable octet. This representation emphasizes the core electrons (those in the Argon core) and explicitly shows the valence electrons responsible for arsenic's chemical behavior. This representation is more precise and reflects the actual energy levels of electrons in arsenic atoms.
Why the Difference Matters
The difference between the simplified and the correct configuration might seem minor, but it's crucial for understanding arsenic's chemical properties. The valence electrons (the outermost electrons in the 4s and 4p orbitals) are responsible for chemical bonding and reactivity. The correct configuration accurately reflects the number and arrangement of these valence electrons, leading to more accurate predictions of arsenic's behavior in chemical reactions.
Delving Deeper into the Exceptions: Why the Aufbau Principle Isn't Always Perfect
The discrepancy between the predicted and actual electron configuration for arsenic highlights an important point: the Aufbau principle is a useful guideline but not an absolute law. The energy levels of orbitals are not always perfectly ordered, and other factors, like electron-electron repulsion and screening effects, can influence the electron arrangement. This is especially true for transition metals and elements like arsenic, which exhibit more complex electron interactions.
The Aufbau principle provides a reasonable approximation, but the actual electronic structure can deviate due to the following factors:
- Shielding Effects: Inner electrons shield the outer electrons from the full positive charge of the nucleus. This shielding reduces the effective nuclear charge experienced by outer electrons, affecting their energy levels.
- Electron-Electron Repulsion: Repulsive forces between electrons can alter the energy levels of orbitals. Electrons in the same subshell will try to maximize their spatial separation to minimize repulsion, potentially influencing the filling order.
- Penetration Effects: Electrons in different orbitals have different probabilities of being close to the nucleus. Electrons in orbitals with better penetration experience a stronger effective nuclear charge and are at lower energy levels.
These factors combine to make the actual energy level ordering more complex than the simple Aufbau principle suggests. Therefore, experimentally determined electron configurations, often obtained via spectroscopic methods, are more accurate than configurations solely derived from the Aufbau principle.
Arsenic's Chemical Properties and its Electron Configuration
The correct electron configuration of arsenic ([Ar] 3d¹⁰ 4s² 4p³) directly influences its chemical properties. The presence of three unpaired electrons in the 4p subshell means arsenic readily forms covalent bonds, often exhibiting a valency of 3 or 5. This explains arsenic's ability to form various compounds and its participation in complex chemical reactions.
Comparing Arsenic's Configuration to its Neighbors
Comparing arsenic's electron configuration to those of its neighboring elements in the periodic table further illustrates the significance of the correct configuration. Consider phosphorus (atomic number 15) and antimony (atomic number 51), both in the same group as arsenic. They too exhibit similar electron configurations with variations in the principal quantum number reflecting their position in the periodic table. Understanding these similarities and differences allows for a deeper understanding of the periodic trends and chemical behaviors of these elements.
Experimental Verification of Arsenic's Electron Configuration
The validity of the [Ar] 3d¹⁰ 4s² 4p³ electron configuration for arsenic isn't just theoretical. Spectroscopic techniques, such as photoelectron spectroscopy (PES) and X-ray photoelectron spectroscopy (XPS), provide experimental evidence supporting this configuration. These techniques can directly probe the energy levels of electrons within an atom, verifying the existence and energies of the 3d, 4s, and 4p orbitals in arsenic atoms and confirming the electron population in each.
Conclusion: Accuracy and Importance in Understanding Arsenic
The accurate electron configuration of arsenic is not just a matter of academic curiosity. It's fundamental to understanding its chemical behavior, reactivity, and bonding patterns. While the Aufbau principle provides a helpful starting point, the influence of electron-electron interactions and the more complex energy level ordering necessitates a more refined approach. The [Ar] 3d¹⁰ 4s² 4p³ configuration accurately reflects these interactions and provides a robust foundation for predicting and explaining arsenic's properties and its role in various chemical and biological systems. This understanding extends beyond arsenic to a broader comprehension of electron configuration exceptions and the importance of experimental verification in refining our models of atomic structure.
Latest Posts
Latest Posts
-
Balanced Equation For Sodium Carbonate And Calcium Chloride
Apr 05, 2025
-
Which Statement Describes Mendels Hypotheses Regarding Gametes
Apr 05, 2025
-
A Perfectly Competitive Firm Is A Price Taker Because
Apr 05, 2025
-
Calculate The Molecular Mass Of H2co3
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Which Is The Correct Electron Configuration For Arsenic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
