Is Dissolving Sugar In Water A Physical Or Chemical Change
News Leon
Apr 02, 2025 · 6 min read
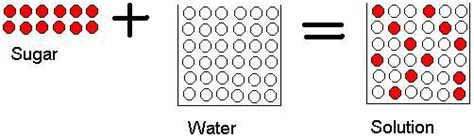
Table of Contents
Is Dissolving Sugar in Water a Physical or Chemical Change? A Deep Dive
The question of whether dissolving sugar in water is a physical or chemical change is a classic introductory chemistry conundrum. While seemingly simple, it delves into the fundamental concepts of matter, its interactions, and the nature of changes it undergoes. Understanding this seemingly simple process provides a crucial foundation for grasping more complex chemical reactions and physical phenomena. This article will explore this question in detail, examining the characteristics of both physical and chemical changes, analyzing the sugar-water interaction, and debunking common misconceptions.
Understanding Physical and Chemical Changes
Before diving into the sugar-water solution, let's clarify the key differences between physical and chemical changes. These distinctions hinge on whether the fundamental nature of the substance involved is altered.
Physical Changes
Physical changes alter the form or appearance of a substance but do not change its chemical composition. These changes are often reversible. Examples include:
- Changes in state: Melting ice (solid to liquid), boiling water (liquid to gas), freezing water (liquid to solid). The chemical composition (H₂O) remains the same.
- Dissolving: Salt dissolving in water. The salt is still salt; it's just dispersed in the water.
- Shape changes: Crushing a can or bending a wire. The metal remains the same chemically.
The key takeaway is that the chemical identity of the substance remains intact after a physical change.
Chemical Changes
Chemical changes, also known as chemical reactions, involve the formation of new substances with different chemical properties. These changes are often irreversible, resulting in a fundamental alteration of the material's composition. Examples include:
- Burning: Wood burning in air forms ash, carbon dioxide, and water. The original wood is no longer present.
- Rusting: Iron reacting with oxygen and water to form iron oxide (rust). The iron's chemical structure is fundamentally altered.
- Cooking an egg: The proteins in the egg undergo chemical changes, leading to irreversible changes in texture and color.
The defining feature of a chemical change is the creation of entirely new substances with different chemical formulas and properties.
Analyzing the Sugar-Water Solution
Now, let's scrutinize the dissolving of sugar in water in light of these definitions. When you add sugar to water and stir, the sugar crystals seemingly disappear, creating a homogeneous solution—a mixture where the components are evenly distributed.
Evidence Suggesting a Physical Change
Several observations strongly suggest that dissolving sugar in water is a physical change:
- Retention of Chemical Identity: The sugar molecules (sucrose, C₁₂H₂₂O₁₁) remain intact throughout the process. No new chemical bonds are formed, and no new chemical substance is created. You could, in theory, recover the sugar by evaporating the water. This reversibility is a hallmark of physical changes.
- No Energy Change (Significant): Dissolving sugar in water might cause a slight temperature change, but this is often negligible and not indicative of a substantial chemical reaction. Chemical reactions often involve significant energy changes (exothermic or endothermic).
- Separation by Physical Means: The sugar and water can be easily separated using simple physical methods, such as evaporation. This further supports the idea that no new chemical bond has formed.
Addressing Potential Arguments for a Chemical Change
While the evidence overwhelmingly points to a physical change, some arguments might initially suggest otherwise:
- Appearance Change: The obvious change in appearance (from solid sugar crystals to a clear solution) could lead to confusion. However, this is merely a change in the physical state and distribution of the sugar, not its chemical composition.
- Interaction of Molecules: Sugar molecules interact with water molecules through intermolecular forces (hydrogen bonds). This interaction, however, doesn't involve the breaking and formation of covalent bonds within the sugar molecules themselves.
The Role of Intermolecular Forces
The dissolving process hinges on the interaction between sugar and water molecules, specifically intermolecular forces. Water is a polar molecule, meaning it has a slightly positive end (hydrogen atoms) and a slightly negative end (oxygen atom). Sucrose, while a larger molecule, also possesses polar regions due to the presence of hydroxyl (-OH) groups.
These polar regions allow sugar molecules to interact with water molecules through hydrogen bonding. Water molecules surround the sugar molecules, effectively pulling them apart from the crystal lattice and dispersing them throughout the solution. This interaction explains the dissolving process without invoking any chemical transformation.
Debunking Common Misconceptions
Several misconceptions often surround the dissolving of sugar in water:
- "Dissolving is always a chemical change": This is incorrect. Dissolving can be either a physical or chemical change, depending on whether the solute undergoes a chemical transformation. Salt dissolving in water is a physical change; certain metals reacting with acid are a chemical change.
- "The change in appearance proves a chemical change": A change in appearance doesn't automatically indicate a chemical change. Many physical changes involve significant alterations in appearance.
- "Any mixture is a chemical change": Mixtures can be created through both physical and chemical processes. The key difference lies in whether the components retain their original chemical identities.
Conclusion: A Physical Change
Based on the evidence presented, dissolving sugar in water is definitively a physical change. The sugar molecules retain their chemical identity throughout the process; no new chemical substances are formed, and the change is easily reversible. While intermolecular forces play a critical role in the dissolution process, these forces don't lead to the breaking or formation of chemical bonds within the sugar or water molecules themselves.
Understanding this distinction is crucial for a solid grasp of fundamental chemistry concepts. It highlights the importance of focusing on the chemical composition of substances before classifying changes as physical or chemical. The apparent simplicity of the sugar-water solution belies its profound significance in illustrating these core principles. This clear understanding forms a robust foundation for exploring more complex chemical and physical phenomena.
Further Exploration: Beyond Sugar and Water
The concept of dissolving extends far beyond sugar and water. Exploring the solubility of various substances in different solvents reveals the fascinating intricacies of intermolecular forces and their influence on physical processes. Investigating factors affecting solubility, such as temperature and pressure, further expands our understanding of these fundamental interactions.
The principles discussed here—the distinction between physical and chemical changes, the role of intermolecular forces, and the importance of identifying changes in chemical composition—are cornerstones of chemistry, applicable to numerous situations and essential to a deeper understanding of the world around us. Continuously challenging our assumptions and rigorously analyzing observations are crucial for acquiring a more comprehensive knowledge of the physical and chemical world.
Latest Posts
Latest Posts
-
Determine Whether The Following Statement Is True Or False
Apr 03, 2025
-
Is C A Metal Or Nonmetal
Apr 03, 2025
-
Is Coal Igneous Sedimentary Or Metamorphic
Apr 03, 2025
-
What Is The Area Of The Shaded Triangle
Apr 03, 2025
-
Which Of The Following Is Not A Secondary Sex Characteristic
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Dissolving Sugar In Water A Physical Or Chemical Change . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
