Is C A Metal Or Nonmetal
News Leon
Apr 03, 2025 · 5 min read
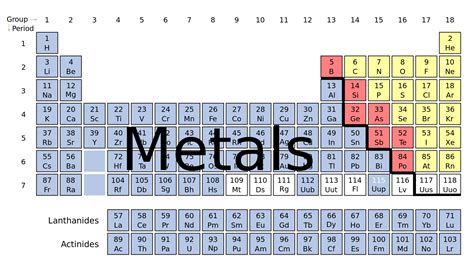
Table of Contents
Is C a Metal or Nonmetal? Unveiling the Secrets of Carbon
Carbon (C), the sixth element on the periodic table, is a fascinating element that defies simple categorization. While many readily identify elements as either metals or nonmetals, carbon presents a unique case, exhibiting properties of both. This article delves deep into the nature of carbon, exploring its various allotropes, chemical behavior, and physical characteristics to definitively answer the question: is carbon a metal or a nonmetal? The short answer is nonmetal, but the nuanced story behind that classification is far more compelling.
Understanding the Metal vs. Nonmetal Divide
Before we classify carbon, let's establish a clear understanding of what distinguishes metals from nonmetals. This distinction is primarily based on their physical and chemical properties.
Metals: A Quick Overview
Metals are typically characterized by:
- High electrical and thermal conductivity: They readily conduct electricity and heat.
- Malleability and ductility: They can be hammered into sheets (malleability) and drawn into wires (ductility).
- Metallic luster: They possess a shiny appearance.
- High density: They are generally dense.
- Tendency to lose electrons: They readily form positive ions (cations) in chemical reactions.
Nonmetals: A Contrasting Profile
Nonmetals, on the other hand, demonstrate:
- Poor electrical and thermal conductivity: They are generally poor conductors of electricity and heat.
- Brittleness: They are often brittle and shatter easily.
- Lack of metallic luster: They typically lack the shiny appearance of metals.
- Lower density: They tend to have lower densities compared to metals.
- Tendency to gain electrons: They readily form negative ions (anions) in chemical reactions.
Carbon's Allotropes: A Tale of Two (and More) Substances
One of the most remarkable aspects of carbon is its ability to exist in various forms called allotropes. These allotropes possess drastically different physical properties despite being composed solely of carbon atoms. The most prominent allotropes are:
1. Diamond: The King of Hardness
Diamond is renowned for its exceptional hardness, making it ideal for industrial cutting and polishing tools, as well as its brilliance as a gemstone. Its structure is a three-dimensional network of strong covalent bonds, creating a rigid, tightly packed structure. This strong bonding accounts for its high hardness and poor electrical conductivity. Clearly, diamond exhibits properties characteristic of nonmetals.
2. Graphite: The Slippery One
Graphite, in stark contrast to diamond, is soft and slippery, used in pencils and as a lubricant. Its structure consists of layers of carbon atoms arranged in hexagonal lattices. These layers are weakly bonded to each other, allowing them to slide easily past one another. This layered structure also accounts for graphite's ability to conduct electricity, a property more typical of metals. However, it's important to note that this conductivity is significantly lower than that of typical metals.
3. Fullerenes: The Spherical Molecules
Fullerenes, such as buckminsterfullerene (C60), also known as "buckyballs," are spherical or ellipsoidal molecules composed of carbon atoms. Their unique structure gives them distinct properties, including potential applications in materials science and medicine. Like diamond, they are generally poor conductors of electricity, aligning with the characteristics of nonmetals.
4. Carbon Nanotubes: Tiny Tubes with Giant Potential
Carbon nanotubes are cylindrical structures made of rolled-up sheets of graphene (a single layer of graphite). These structures exhibit exceptional strength and electrical conductivity, depending on their structure. Some nanotubes can conduct electricity even better than copper, a quintessential metal. However, this exceptional conductivity is specific to the nanotube's structure and arrangement, not a general property of all forms of carbon.
5. Amorphous Carbon: The Unstructured Form
Amorphous carbon lacks the ordered crystalline structure of diamond and graphite. It's a more disordered form found in materials like coal and soot. Its properties vary widely depending on its specific preparation and structure, but it generally displays the properties of a nonmetal, with poor conductivity and brittleness.
The Chemical Behavior of Carbon: A Nonmetal's Tale
Beyond its physical properties, carbon's chemical behavior further solidifies its classification as a nonmetal. Carbon's electronegativity, a measure of an atom's ability to attract electrons in a chemical bond, is relatively high. This means carbon tends to share electrons with other atoms to form covalent bonds, rather than readily losing electrons to form positive ions like metals do. This covalent bonding is a hallmark characteristic of nonmetals.
Carbon's ability to form long chains and rings of carbon atoms is also a crucial aspect of its chemistry. This property, known as catenation, underpins the vast diversity of organic compounds, which form the basis of life itself. This complexity and tendency for covalent bonding are strong indicators that it is a nonmetal.
The Ambiguity: Graphite's Conductivity
The electrical conductivity of graphite presents a potential challenge to definitively labeling carbon as a nonmetal. However, it is crucial to remember that graphite’s conductivity is due to its unique layered structure and the delocalized electrons within those layers. This isn't the same mechanism as the sea of delocalized electrons found in metallic bonds, responsible for the high conductivity in typical metals. The conductivity of graphite is still significantly lower than that of most metals.
Conclusion: Carbon - A Nonmetal with Exceptional Versatility
While the existence of allotropes like graphite displays some metallic-like properties such as conductivity, the overwhelming evidence points to carbon being a nonmetal. Its chemical behavior, dominated by covalent bonding and high electronegativity; its tendency to form long chains and rings (catenation); and the properties of its major allotropes (diamond, fullerenes, amorphous carbon) strongly suggest a nonmetallic classification. The exceptional properties of carbon nanotubes, showcasing superior conductivity, are a result of their highly specific and engineered structure, not an inherent characteristic of the element itself. Therefore, despite the intriguing diversity of its allotropes, carbon's overarching properties firmly place it in the realm of nonmetals. This, however, does not diminish its importance or the extraordinary diversity of its applications in various fields. Its unique properties continue to inspire innovation and discoveries in materials science, chemistry, and beyond.
Latest Posts
Latest Posts
-
Is The Human Body A Conductor
Apr 04, 2025
-
80 Of 45 Is What Number
Apr 04, 2025
-
What Is The Greatest Common Factor Of 36 And 84
Apr 04, 2025
-
What Type Of Triangle Is Shown
Apr 04, 2025
-
Curdling Of Milk Is A Chemical Change
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Is C A Metal Or Nonmetal . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
