How To Draw A Carbon Atom
News Leon
Apr 05, 2025 · 6 min read
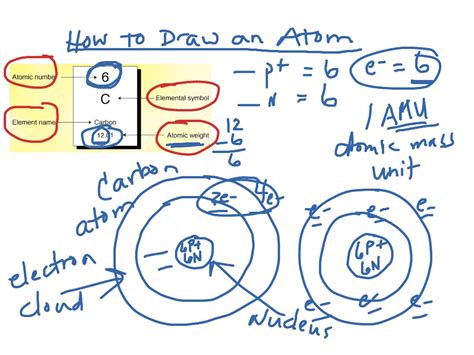
Table of Contents
How to Draw a Carbon Atom: A Comprehensive Guide for Beginners and Beyond
Drawing a carbon atom might seem deceptively simple, but accurately representing its structure and properties requires understanding fundamental chemistry concepts. This comprehensive guide will walk you through various methods of drawing a carbon atom, from basic Lewis dot structures to more advanced representations used in organic chemistry and beyond. We'll cover everything from the fundamental principles to nuanced details, ensuring you can confidently depict this fundamental building block of life and countless other molecules.
Understanding the Basics: What Makes Carbon Special?
Before diving into drawing techniques, let's establish the core properties of a carbon atom that influence how we represent it visually:
- Atomic Number: Carbon (C) has an atomic number of 6, meaning it possesses six protons and six electrons. This electron configuration is crucial for its bonding capabilities.
- Electron Configuration: The electron configuration of carbon is 1s²2s²2p². This means two electrons occupy the innermost shell (1s), and four electrons occupy the outer shell (2s and 2p). These four outer electrons are valence electrons, responsible for carbon's ability to form up to four covalent bonds.
- Covalent Bonding: Carbon predominantly forms covalent bonds, sharing electrons with other atoms to achieve a stable octet (eight electrons in its outermost shell). This bonding characteristic is pivotal to the vast diversity of organic molecules.
Method 1: The Simple Lewis Dot Structure
The simplest way to represent a carbon atom is using a Lewis dot structure. This method visually depicts the valence electrons:
- The Symbol: Start with the symbol "C" representing the carbon atom.
- Valence Electrons: Place four dots around the symbol, representing the four valence electrons. These dots should be evenly spaced, typically one on each side of the symbol.
.
. C .
.
This basic representation emphasizes carbon's four valence electrons and its potential to form four bonds. While simple, it doesn't show the electron orbitals or the three-dimensional nature of the atom.
Method 2: Representing Electron Orbitals (Bohr Model)
A slightly more advanced representation incorporates the concept of electron orbitals. The Bohr model, while simplified, helps visualize electron shells:
- The Nucleus: Draw a small circle in the center representing the nucleus, containing six protons and six neutrons (for carbon-12, the most common isotope).
- Electron Shells: Draw two concentric circles around the nucleus. The inner circle represents the first electron shell (1s), containing two electrons. The outer circle represents the second electron shell (2s and 2p), containing four electrons.
- Electron Placement: Place two electrons in the inner shell and four electrons in the outer shell. It's important to note that the Bohr model doesn't accurately depict the shapes of orbitals but serves as a useful visualization.
This method provides a better understanding of electron arrangement within the atom, but it still lacks the accurate representation of orbital shapes.
Method 3: Showing Orbital Hybridization (sp³, sp², sp)
To accurately represent carbon's bonding capabilities, we need to consider orbital hybridization. Carbon's ability to form diverse structures arises from the hybridization of its atomic orbitals:
-
sp³ Hybridization (Tetrahedral): In methane (CH₄), carbon's 2s and three 2p orbitals hybridize to form four equivalent sp³ orbitals. These orbitals are arranged tetrahedrally, creating a three-dimensional structure with bond angles of approximately 109.5°. To represent this:
- The Central Carbon: Draw a central carbon atom.
- Tetrahedral Arrangement: Draw four lines extending from the carbon atom, each representing a covalent bond to another atom (e.g., hydrogen in methane). These lines should be spatially distributed to reflect the tetrahedral geometry.
- Bond Angles: While not always explicitly shown, understanding the approximate 109.5° bond angle is crucial.
-
sp² Hybridization (Trigonal Planar): In molecules like ethene (C₂H₄), carbon undergoes sp² hybridization, forming three equivalent sp² orbitals and one unhybridized p orbital. The sp² orbitals are arranged in a trigonal planar geometry (120° bond angles). This unhybridized p orbital participates in a pi bond. Representation would involve:
- Central Carbon: Draw the central carbon atom.
- Trigonal Planar Geometry: Draw three lines extending from the carbon, representing bonds to other atoms, arranged at 120° angles.
- Pi Bond (Optional): The unhybridized p orbital can be shown as a sideways overlapping orbital.
-
sp Hybridization (Linear): In molecules like ethyne (C₂H₂), carbon undergoes sp hybridization, forming two sp orbitals and two unhybridized p orbitals. The sp orbitals are linearly arranged (180° bond angles). Representation involves:
- Central Carbon: Draw the central carbon atom.
- Linear Arrangement: Draw two lines extending from the carbon at a 180° angle.
- Pi Bonds (Optional): The two unhybridized p orbitals can be shown as two pairs of overlapping orbitals.
Method 4: Ball-and-Stick Models and Space-Filling Models
Three-dimensional models provide the most realistic representations of carbon atoms within molecules. These models often use different approaches:
- Ball-and-Stick Models: These models use balls to represent atoms and sticks to represent bonds. The size of the balls can be proportional to atomic radii. They clearly show the connectivity and bond angles.
- Space-Filling Models: These models use spheres to represent atoms whose sizes reflect their van der Waals radii. They provide a better representation of the molecule's overall shape and volume, emphasizing the spatial arrangement of atoms and avoiding gaps.
These 3D models are especially useful for visualizing complex organic molecules and understanding steric hindrance and other spatial relationships within a molecule.
Advanced Representations: Beyond Basic Drawings
As you delve deeper into chemistry, you'll encounter more advanced ways to represent carbon atoms and molecules:
- Skeletal Formulae: In organic chemistry, skeletal formulae are used to simplify the depiction of organic molecules. Carbon atoms are implied at the intersection of lines and at the end of lines. Hydrogen atoms bonded to carbon are usually omitted for brevity.
- Condensed Formulae: These formulae represent molecules by listing the atoms and their connections in a linear format, grouping atoms bonded to the same carbon together.
- SMILES Notation: SMILES (Simplified Molecular Input Line Entry System) is a widely used chemical notation for representing molecules as strings of characters. It's particularly useful for computer-based applications in cheminformatics.
Practical Exercises for Drawing Carbon Atoms
To solidify your understanding, try these exercises:
- Draw a Lewis dot structure for a carbon atom.
- Draw a Bohr model of a carbon atom.
- Draw a methane molecule (CH₄) using sp³ hybridization. Clearly show the tetrahedral geometry and bond angles.
- Draw an ethene molecule (C₂H₄) using sp² hybridization. Indicate the sigma and pi bonds.
- Draw an ethyne molecule (C₂H₂) using sp hybridization. Indicate the sigma and pi bonds.
- Draw a ball-and-stick model of methane (CH₄).
- Draw a space-filling model of methane (CH₄).
- Draw the skeletal formula for butane (C₄H₁₀).
By completing these exercises, you'll gain a practical understanding of how to represent a carbon atom in different contexts and levels of detail.
Conclusion: Mastering the Art of Drawing Carbon Atoms
Drawing carbon atoms effectively requires understanding its electronic structure and bonding properties. From simple Lewis dot structures to complex 3D models, the chosen method depends on the context and desired level of detail. By mastering these techniques, you'll be well-equipped to represent this fundamental element and its role in the vast world of organic and inorganic chemistry. Remember to practice regularly to enhance your skills and deepen your understanding of this essential building block of the universe.
Latest Posts
Latest Posts
-
A Pendulum Consists Of A Small Object
Apr 06, 2025
-
Fertilization In Humans Normally Occurs In The
Apr 06, 2025
-
What Is The Ph Of A 0 001 M Koh Solution
Apr 06, 2025
-
What Is 0 6 As A Fraction In Simplest Form
Apr 06, 2025
-
Write Iupac Name Of The Following Compound
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How To Draw A Carbon Atom . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
