How Many Unpaired Electrons Does A Sulfur Atom Have
News Leon
Apr 04, 2025 · 5 min read
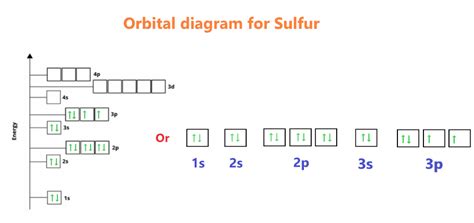
Table of Contents
How Many Unpaired Electrons Does a Sulfur Atom Have? A Deep Dive into Atomic Structure and Electron Configuration
Understanding the number of unpaired electrons in a sulfur atom requires a journey into the fascinating world of atomic structure and electron configuration. This seemingly simple question opens the door to a deeper appreciation of how electrons arrange themselves within an atom, influencing its chemical properties and reactivity. Let's embark on this journey, exploring the fundamental principles and arriving at a definitive answer.
Diving into Atomic Structure: The Basics
Before we delve into the specifics of sulfur, let's establish a foundational understanding of atomic structure. Atoms, the fundamental building blocks of matter, are composed of three primary subatomic particles:
- Protons: Positively charged particles residing in the atom's nucleus. The number of protons defines the element's atomic number and its identity.
- Neutrons: Neutrally charged particles also located in the nucleus. The number of neutrons can vary within the same element, resulting in isotopes.
- Electrons: Negatively charged particles orbiting the nucleus in specific energy levels or shells. These electrons are crucial for determining an atom's chemical behavior.
It's the arrangement of these electrons, specifically their distribution within the electron shells and subshells, that dictates an atom's properties and its ability to form chemical bonds.
Electron Configuration: The Key to Understanding Electron Pairing
The electron configuration of an atom describes how its electrons are distributed among various energy levels and sublevels. This arrangement follows specific rules, primarily governed by the Aufbau principle (electrons fill lower energy levels first), Hund's rule (electrons occupy orbitals individually before pairing up), and the Pauli exclusion principle (each orbital can hold a maximum of two electrons with opposite spins).
Understanding these rules is crucial for determining the number of unpaired electrons. Let's break down each rule:
-
Aufbau Principle: Electrons fill orbitals starting with the lowest energy level. The order of filling is typically: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on.
-
Hund's Rule: Within a subshell (like the p subshell with three orbitals), electrons will individually occupy each orbital before pairing up in any one orbital. This minimizes electron-electron repulsion.
-
Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons, but these two electrons must have opposite spins (represented as ↑ and ↓).
Determining the Electron Configuration of Sulfur (S)
Sulfur has an atomic number of 16, meaning it has 16 protons and, in a neutral atom, 16 electrons. Using the Aufbau principle, we can determine its electron configuration:
1s² 2s² 2p⁶ 3s² 3p⁴
Let's break this down:
- 1s²: The first energy level (n=1) contains the 's' subshell, which can hold up to two electrons. Sulfur has two electrons in this subshell.
- 2s²: The second energy level (n=2) also contains an 's' subshell with two electrons.
- 2p⁶: The second energy level also contains a 'p' subshell, which has three orbitals and can hold up to six electrons. Sulfur fills this subshell completely.
- 3s²: The third energy level (n=3) begins with an 's' subshell holding two electrons.
- 3p⁴: The third energy level's 'p' subshell contains four electrons.
Unveiling the Unpaired Electrons in Sulfur
Now comes the crucial step: analyzing the 3p subshell. Remember Hund's rule! The 3p subshell has three orbitals, each capable of holding two electrons. With four electrons to distribute, we get:
↑ ↑ ↑
Each arrow represents an electron with a specific spin. Notice that two orbitals are singly occupied, and one orbital is doubly occupied.
Therefore, sulfur (S) has two unpaired electrons.
The Significance of Unpaired Electrons
The presence of unpaired electrons significantly influences an atom's properties:
-
Paramagnetism: Atoms with unpaired electrons are paramagnetic, meaning they are weakly attracted to a magnetic field. This is because the unpaired electrons' spins create a small magnetic moment.
-
Chemical Reactivity: Unpaired electrons are highly reactive, readily participating in chemical bond formation. Sulfur's two unpaired electrons contribute to its ability to form various compounds, often with oxidation states of -2, +4, or +6. It readily forms covalent bonds by sharing these electrons with other atoms.
Beyond the Basics: Excited States and Exceptions
While the ground state electron configuration discussed above is the most stable arrangement, sulfur can exist in excited states under specific conditions (like absorbing energy). In these excited states, an electron might jump to a higher energy level, potentially altering the number of unpaired electrons. However, the ground state configuration is the most relevant for understanding sulfur's typical chemical behavior.
Sulfur's Role in the World Around Us
Understanding sulfur's atomic structure and its two unpaired electrons is crucial for appreciating its importance in various fields:
-
Biological Systems: Sulfur is an essential component of many proteins and enzymes, playing a critical role in biological processes. The cysteine amino acid, containing a sulfur atom, forms disulfide bonds which are essential for protein structure and function.
-
Industrial Applications: Sulfur is a crucial element in the production of sulfuric acid, a widely used industrial chemical with applications ranging from fertilizer production to the refining of petroleum.
-
Environmental Science: Sulfur compounds play a significant role in atmospheric chemistry and contribute to acid rain formation. Understanding sulfur's reactivity is crucial for mitigating environmental pollution.
Conclusion: The Importance of Electron Configuration
Determining the number of unpaired electrons in a sulfur atom – two, in its ground state – is more than just a simple exercise. It provides a gateway to understanding the fundamental principles of atomic structure and electron configuration. This knowledge allows us to predict an element's chemical properties, its reactivity, and its role in the world around us. The seemingly simple question of unpaired electrons opens a window into the complex and fascinating world of chemistry. By understanding these fundamental concepts, we can appreciate the intricate relationships between atomic structure, electron configuration, and the macroscopic properties of matter. This deeper understanding allows us to explore and innovate in various scientific and technological fields.
Latest Posts
Latest Posts
-
What Is The Formula For Magnesium Carbonate
Apr 05, 2025
-
Complete The Curved Arrow Mechanism Of The Following Double Elimination
Apr 05, 2025
-
A Wave That Does Not Require A Medium To Travel
Apr 05, 2025
-
Provides The Instructions That Tell A Computer What To Do
Apr 05, 2025
-
Amino Acids And Glucose Are Reabsorbed Primarily In The
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Many Unpaired Electrons Does A Sulfur Atom Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
