How Many Protons Neutrons And Electrons In Iron
News Leon
Apr 03, 2025 · 6 min read
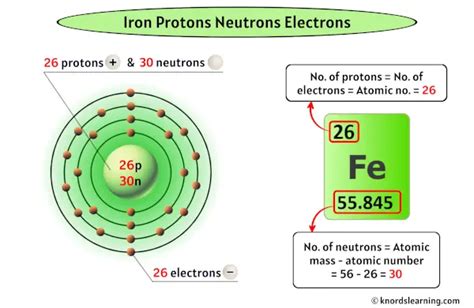
Table of Contents
How Many Protons, Neutrons, and Electrons in Iron? A Deep Dive into Atomic Structure
Iron, a ubiquitous element crucial to life and industry, presents a fascinating case study in atomic structure. Understanding its composition – the number of protons, neutrons, and electrons – unlocks insights into its properties and behavior. This comprehensive guide delves into the specifics of iron's atomic makeup, exploring its isotopes, applications, and significance within the broader context of chemistry and physics.
Understanding Atomic Structure: The Building Blocks of Matter
Before we delve into the specifics of iron, let's establish a foundational understanding of atomic structure. All matter is composed of atoms, the fundamental units of chemical elements. Each atom consists of three subatomic particles:
- Protons: Positively charged particles residing in the atom's nucleus. The number of protons defines the element's atomic number and determines its identity.
- Neutrons: Neutral particles (no charge) also found within the nucleus. They contribute to the atom's mass but not its charge.
- Electrons: Negatively charged particles orbiting the nucleus in electron shells or energy levels. The number of electrons usually equals the number of protons in a neutral atom.
Iron's Atomic Number and Protons: Defining the Element
Iron's atomic number is 26. This crucial number signifies that every iron atom contains 26 protons. This is non-negotiable; the presence of 26 protons is the defining characteristic that distinguishes iron from all other elements. Changing the number of protons fundamentally changes the element.
Iron's Neutrons: Isotopes and Atomic Mass
Unlike the fixed number of protons, the number of neutrons in an iron atom can vary. Atoms of the same element with differing neutron counts are called isotopes. Iron has several naturally occurring isotopes, each with a different atomic mass. Atomic mass is the sum of the number of protons and neutrons.
The most common isotopes of iron are:
- Iron-56 (⁵⁶Fe): This is the most abundant isotope, constituting about 91.75% of naturally occurring iron. It has 26 protons and 30 neutrons (26 + 30 = 56).
- Iron-54 (⁵⁴Fe): This isotope makes up approximately 5.85% of natural iron. It contains 26 protons and 28 neutrons.
- Iron-57 (⁵⁷Fe): Present in about 2.12% of natural iron, this isotope has 26 protons and 31 neutrons.
- Iron-58 (⁵⁸Fe): A less abundant isotope comprising approximately 0.28% of natural iron. It has 26 protons and 32 neutrons.
These are just the most common isotopes. Other, less abundant isotopes of iron exist, both stable and unstable (radioactive). The average atomic mass of iron, often listed on periodic tables, is a weighted average of the masses of its naturally occurring isotopes, taking into account their relative abundances. This weighted average is approximately 55.845 atomic mass units (amu).
Iron's Electrons: Charge Neutrality and Chemical Behavior
In a neutral iron atom, the number of electrons precisely matches the number of protons. Therefore, a neutral iron atom has 26 electrons. These electrons are arranged in shells or energy levels surrounding the nucleus, following specific rules dictated by quantum mechanics. The electron configuration of iron is [Ar] 3d⁶ 4s². This electron arrangement significantly influences iron's chemical reactivity and its ability to form various chemical bonds.
The distribution of electrons in the outermost shell (valence electrons) determines an element's chemical behavior. Iron's valence electrons participate in chemical reactions, allowing it to form compounds with other elements. This explains iron's versatility and its presence in countless compounds and materials.
The Significance of Iron's Atomic Structure in its Properties
Iron's unique atomic structure accounts for many of its remarkable properties, making it a pivotal element in various fields:
- Strength and Durability: The strong metallic bonds between iron atoms result from the interaction of their valence electrons. This explains iron's strength, making it a structural material in buildings, vehicles, and infrastructure.
- Magnetic Properties: The specific arrangement of electrons in iron's d orbitals leads to its ferromagnetic properties. This means that iron can be easily magnetized and retain its magnetism, a property exploited in countless applications.
- Reactivity: Iron's ability to readily react with oxygen and other elements is a double-edged sword. While it contributes to the formation of essential compounds, it also leads to corrosion and rust formation.
- Biological Importance: Iron is essential for life. It's a component of hemoglobin, the protein in red blood cells that carries oxygen throughout the body. Its role in various biological processes highlights its critical significance in living organisms.
- Industrial Applications: Iron's properties have led to its widespread use in numerous industries, including construction, manufacturing, and transportation. Steel, an alloy of iron and carbon, is one of the most widely used materials globally.
Isotopes and Applications: Beyond the Basics
The different isotopes of iron, while possessing the same number of protons, exhibit subtle variations in their properties. These variations have implications in various fields:
- Iron-57 in Mössbauer Spectroscopy: This isotope is crucial in Mössbauer spectroscopy, a technique used to study the properties of materials and their interactions. Its unique nuclear properties make it an ideal probe for investigating magnetic and structural properties.
- Iron-59 as a Tracer: This radioactive isotope finds application as a tracer in medical and biological studies to track iron's movement and metabolism within the body.
- Iron Isotope Analysis in Geology and Archaeology: Variations in the relative abundances of iron isotopes in geological samples and artifacts can provide insights into past geological processes and historical events.
Beyond the Basics: Nuclear Physics and Iron
Delving deeper, the nuclear stability of iron isotopes is a significant area of study in nuclear physics. Iron-56, the most abundant isotope, represents a local minimum in the curve of binding energy per nucleon. This means that it is exceptionally stable compared to other nearby nuclei, contributing to its abundance in the universe. Understanding the processes that lead to the formation and abundance of iron isotopes is essential for cosmology and nuclear astrophysics. Supernova explosions, for instance, are a significant source of iron in the universe, producing heavier elements through nucleosynthesis.
Conclusion: Iron's Enduring Importance
The seemingly simple answer to "how many protons, neutrons, and electrons are in iron?" opens a door to a fascinating world of atomic structure, chemical properties, and industrial applications. The number of protons (26) defines iron as an element, while variations in neutrons give rise to its isotopes, each with unique characteristics and applications. The 26 electrons determine iron's chemical reactivity and its role in biological systems and industrial processes. Understanding iron's atomic structure offers a crucial window into the nature of matter and its importance across diverse scientific fields. From the building blocks of life to the construction of skyscrapers, iron's influence is undeniable, a testament to the profound impact of its unique atomic composition.
Latest Posts
Latest Posts
-
An Interior Angle Of A Regular Polygon Measures 170
Apr 04, 2025
-
A Fluids Resistance To Flow Is Called
Apr 04, 2025
-
Match The Location With The Appropriate Epithelial Tissue
Apr 04, 2025
-
Sodium Sulfate Barium Chloride Balanced Equation
Apr 04, 2025
-
Which Of The Following Compounds Has The Lowest Boiling Point
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Many Protons Neutrons And Electrons In Iron . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
