Give The Iupac Names For The Following Compounds
News Leon
Apr 03, 2025 · 6 min read
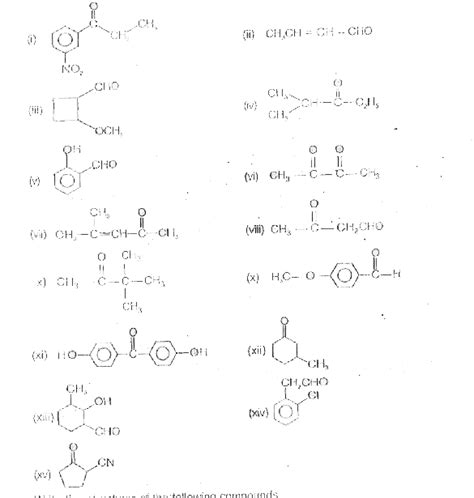
Table of Contents
Giving IUPAC Names to Organic Compounds: A Comprehensive Guide
Naming organic compounds might seem daunting at first, but with a systematic approach and understanding of IUPAC nomenclature rules, it becomes a straightforward process. The International Union of Pure and Applied Chemistry (IUPAC) has established a standardized system for naming organic compounds, ensuring clear and unambiguous communication among chemists worldwide. This guide provides a comprehensive walkthrough of how to name various organic compounds, covering alkanes, alkenes, alkynes, alcohols, aldehydes, ketones, carboxylic acids, and more, accompanied by numerous examples.
Understanding the Basics of IUPAC Nomenclature
The foundation of IUPAC nomenclature lies in identifying the parent chain, functional groups, and substituents.
-
Parent Chain: This is the longest continuous carbon chain in the molecule. The name of the parent chain determines the base name of the compound. For example, a chain of five carbons is a pentane, six carbons is hexane, and so on.
-
Functional Groups: These are specific groups of atoms within a molecule that confer characteristic chemical properties. Examples include hydroxyl (-OH) groups in alcohols, carbonyl (C=O) groups in aldehydes and ketones, and carboxyl (-COOH) groups in carboxylic acids. The presence of a functional group significantly influences the naming process.
-
Substituents: These are atoms or groups of atoms attached to the parent chain that are not part of the main functional group. They are named as prefixes to the parent chain name. Common substituents include alkyl groups (e.g., methyl, ethyl, propyl) and halogens (e.g., chloro, bromo, iodo).
Naming Alkanes: The Foundation
Alkanes are saturated hydrocarbons, meaning they contain only single bonds between carbon atoms. The naming of alkanes forms the basis for naming many other organic compounds.
-
Straight-chain alkanes: These are named using prefixes indicating the number of carbon atoms (meth- for 1, eth- for 2, prop- for 3, but- for 4, pent- for 5, hex- for 6, hept- for 7, oct- for 8, non- for 9, dec- for 10, and so on) followed by the suffix "-ane".
-
Branched-chain alkanes: For branched alkanes, the longest continuous carbon chain is identified as the parent chain. The substituents (alkyl groups) are named and numbered according to their position on the parent chain. The numbering starts from the end that gives the substituents the lowest possible numbers. If multiple substituents are present, they are listed alphabetically, ignoring prefixes like "di-", "tri-", etc., unless these prefixes are part of the substituent's name (e.g., isopropyl).
Examples:
- CH₃CH₂CH₂CH₃: Butane
- CH₃CH(CH₃)CH₃: 2-Methylpropane
- CH₃CH₂CH(CH₃)CH₂CH₃: 3-Methylpentane
- CH₃CH(CH₃)CH₂CH(CH₃)CH₃: 2,4-Dimethylpentane
- CH₃C(CH₃)₂CH₂CH₃: 2,2-Dimethylbutane
Naming Alkenes and Alkynes
Alkenes contain at least one carbon-carbon double bond, while alkynes contain at least one carbon-carbon triple bond.
-
Alkenes: The longest chain containing the double bond is the parent chain. The suffix "-ene" replaces the "-ane" of the corresponding alkane. The position of the double bond is indicated by the lowest possible number.
-
Alkynes: Similar to alkenes, the longest chain containing the triple bond is the parent chain. The suffix "-yne" replaces the "-ane". The position of the triple bond is indicated by the lowest possible number.
Examples:
- CH₂=CHCH₂CH₃: 1-Butene
- CH₃CH=CHCH₃: 2-Butene
- CH≡CCH₂CH₃: 1-Butyne
- CH₃C≡CCH₃: 2-Butyne
- CH₃CH=CHCH₂CH=CH₂: 1,4-Hexadiene
Naming Alcohols, Aldehydes, and Ketones
These compound classes contain oxygen-containing functional groups.
-
Alcohols: The -OH (hydroxyl) group is the characteristic functional group. The suffix "-ol" is added to the parent alkane name, and the position of the hydroxyl group is indicated by a number.
-
Aldehydes: The -CHO (aldehyde) group is always located at the end of the carbon chain. The suffix "-al" is used.
-
Ketones: The -CO- (carbonyl) group is located within the carbon chain. The suffix "-one" is used, and the position of the carbonyl group is indicated by a number.
Examples:
- CH₃CH₂OH: Ethanol
- CH₃CH(OH)CH₃: 2-Propanol
- CH₃CHO: Ethanal
- CH₃COCH₃: Propanone (Acetone)
- CH₃CH₂COCH₃: Butanone
Naming Carboxylic Acids, Esters, and Amines
These compound classes represent other crucial functional groups.
-
Carboxylic Acids: The -COOH (carboxyl) group is the characteristic functional group, always at the end of the chain. The suffix "-oic acid" is used. The carbon of the carboxyl group is always carbon number 1.
-
Esters: Esters are derived from carboxylic acids. They are named by first naming the alkyl group attached to the oxygen atom, followed by the name of the carboxylate anion (derived from the carboxylic acid).
-
Amines: Amines contain a nitrogen atom bonded to one or more alkyl groups. They are named by identifying the alkyl groups attached to the nitrogen, using prefixes like "N-" to indicate substitution on the nitrogen atom itself.
Examples:
- CH₃COOH: Ethanoic acid (Acetic acid)
- CH₃CH₂COOH: Propanoic acid
- CH₃COOCH₂CH₃: Ethyl ethanoate (Ethyl acetate)
- CH₃COOCH₃: Methyl ethanoate (Methyl acetate)
- CH₃NH₂: Methanamine
- CH₃CH₂NHCH₃: N-Methylethanamine
Handling Multiple Functional Groups and Complex Structures
When a molecule contains multiple functional groups, the priority of the functional groups determines the base name and suffixes. The IUPAC rules specify a hierarchy of functional groups, with some taking precedence over others. Carboxylic acids have the highest priority, followed by aldehydes, ketones, alcohols, amines, and so on.
In complex structures with multiple substituents and branches, meticulous numbering and alphabetization are crucial. Remember to always choose the numbering that gives the lowest numbers to the substituents and functional groups.
Advanced Nomenclature Considerations
-
Cyclic Compounds: Cyclic compounds, such as cyclohexane, cyclopentane, require specific naming rules incorporating the prefix "cyclo-" before the alkane name. Substituents on the ring are numbered to provide the lowest possible number set.
-
Stereochemistry: The IUPAC system also incorporates descriptors for stereochemistry (e.g., cis, trans, R, S configurations) to provide a complete description of the molecule's three-dimensional structure. These prefixes are added before the base name to specify the stereochemistry at specific chiral centers or double bonds.
-
Polyfunctional Compounds: For compounds with multiple functional groups of equal or similar priority, the naming becomes more complex, and specific rules within the IUPAC guidelines must be followed. Often, the most oxidized carbon-containing group takes priority.
Practice Makes Perfect
Mastering IUPAC nomenclature requires practice. Start with simple examples and gradually progress to more complex molecules. Work through numerous examples and use online resources and textbooks to reinforce your understanding. The key is to systematically identify the parent chain, functional groups, and substituents, and then apply the appropriate naming rules. Regular practice and a methodical approach will solidify your skills and enable you to confidently name a wide range of organic compounds. By consistently following the established rules, you ensure unambiguous communication and avoid potential confusion in chemical descriptions. Remember, precise naming is fundamental to effective communication within the field of chemistry.
Latest Posts
Latest Posts
-
When A Nerve Fiber Is Polarized The Concentration Of
Apr 04, 2025
-
Which Of The Following Statements Is True Regarding
Apr 04, 2025
-
Is Drawings A Debit Or Credit
Apr 04, 2025
-
Enzymes Are Catalysts That Increase The Rate Of Reactions By
Apr 04, 2025
-
Does Nitrogen Lose Or Gain Electrons
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Give The Iupac Names For The Following Compounds . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
