Enter The Formula For The Compound Barium Oxide.
News Leon
Apr 06, 2025 · 5 min read
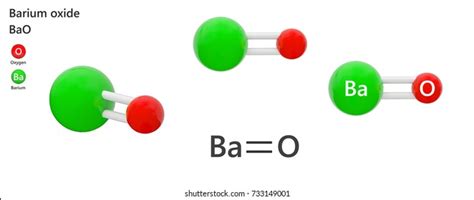
Table of Contents
Enter the Formula for the Compound Barium Oxide
Barium oxide, a fascinating inorganic compound, holds a significant place in various chemical and industrial applications. Understanding its chemical formula, properties, and uses is crucial for anyone working with this substance. This comprehensive guide delves into the intricacies of barium oxide, exploring its formula, synthesis, properties, safety considerations, and diverse applications.
Understanding the Chemical Formula of Barium Oxide
The chemical formula for barium oxide is BaO. This simple formula succinctly represents the compound's composition: one barium (Ba) atom bonded to one oxygen (O) atom. This ionic bond forms due to the significant electronegativity difference between barium, an alkaline earth metal, and oxygen, a highly electronegative nonmetal. Barium readily loses its two valence electrons to achieve a stable electron configuration, while oxygen gains those two electrons to complete its octet. This electron transfer results in the formation of the Ba²⁺ cation and the O²⁻ anion, held together by electrostatic attraction.
Barium's Role in the Compound
Barium (Ba), belonging to Group 2 of the periodic table, is an alkaline earth metal characterized by its high reactivity. Its tendency to lose two electrons to achieve a stable noble gas configuration (like Xenon) is a driving force in its chemical interactions. In barium oxide, barium's +2 oxidation state is crucial for the formation of the stable ionic bond with oxygen.
Oxygen's Contribution to Barium Oxide
Oxygen (O), a highly reactive nonmetal, plays a crucial role in the formation of barium oxide. Its high electronegativity makes it readily accept electrons from other atoms, including barium. In barium oxide, oxygen exists as the oxide anion (O²⁻), completing its electron shell and attaining a stable octet configuration.
Synthesis of Barium Oxide
Several methods exist for synthesizing barium oxide. The most common method involves the direct reaction of barium metal with oxygen gas.
Direct Reaction of Barium with Oxygen
This method, represented by the balanced chemical equation below, provides a straightforward synthesis route:
2Ba(s) + O₂(g) → 2BaO(s)
This reaction is highly exothermic, releasing a significant amount of heat. It requires careful control of reaction conditions to prevent uncontrolled reactions and ensure the formation of pure barium oxide.
Thermal Decomposition of Barium Carbonate
Another method involves the thermal decomposition of barium carbonate (BaCO₃). Heating barium carbonate to high temperatures (above 1360°C) drives off carbon dioxide (CO₂), leaving behind barium oxide:
BaCO₃(s) → BaO(s) + CO₂(g)
This method requires precise temperature control to ensure complete decomposition and prevent the formation of unwanted byproducts.
Physical and Chemical Properties of Barium Oxide
Barium oxide possesses several distinctive physical and chemical properties that contribute to its various applications.
Physical Properties
- Appearance: Barium oxide is a white to yellowish-white crystalline solid.
- Melting Point: It has a relatively high melting point, around 1923°C.
- Boiling Point: Its boiling point is even higher, exceeding 2000°C.
- Solubility: Barium oxide is sparingly soluble in water, reacting to form barium hydroxide (Ba(OH)₂).
- Density: It has a density higher than water.
Chemical Properties
- Reactivity with Water: Barium oxide reacts vigorously with water to form barium hydroxide, releasing significant heat:
BaO(s) + H₂O(l) → Ba(OH)₂(aq)
This reaction is highly exothermic and should be handled with caution.
- Reaction with Acids: Barium oxide readily reacts with acids to form the corresponding barium salts. For example, its reaction with hydrochloric acid (HCl) produces barium chloride (BaCl₂):
BaO(s) + 2HCl(aq) → BaCl₂(aq) + H₂O(l)
- Reaction with Carbon Dioxide: Barium oxide readily absorbs carbon dioxide from the air, forming barium carbonate:
BaO(s) + CO₂(g) → BaCO₃(s)
This property makes it useful as a drying agent in certain applications.
Safety Precautions when Handling Barium Oxide
Barium oxide is considered toxic and should be handled with appropriate safety measures. Direct contact with skin or eyes can cause irritation, and ingestion can lead to serious health issues. Therefore, it's crucial to:
- Wear appropriate personal protective equipment (PPE): This includes gloves, safety goggles, and a lab coat to minimize direct contact with the substance.
- Work in a well-ventilated area: To avoid inhaling dust particles, ensure adequate ventilation during handling and processing.
- Avoid ingestion: Never consume barium oxide, and always wash hands thoroughly after handling.
- Store properly: Keep barium oxide in a tightly sealed container away from moisture and sources of ignition.
Applications of Barium Oxide
Barium oxide finds applications in various fields due to its unique properties.
Use in Glass Manufacturing
Barium oxide acts as a flux in glass manufacturing, lowering the melting point of the glass mixture and improving its properties. It also enhances the refractive index and contributes to the overall quality of the glass.
Application in Ceramics
Similar to its role in glass production, barium oxide contributes to the properties of ceramics. It enhances their durability and resistance to heat and chemicals.
Use as a Catalyst
Barium oxide serves as a catalyst in various chemical reactions, accelerating the rate of reactions without being consumed in the process.
Role in Electronic Devices
Barium oxide is used in the production of certain electronic components due to its electrical conductivity and other unique properties.
Use in Metallurgy
In the metallurgy industry, barium oxide plays a role in metal refining and alloy production.
Miscellaneous Applications
Besides these major applications, barium oxide finds smaller-scale uses in other areas, including the production of certain pigments and as a component in various chemical formulations.
Future Prospects of Barium Oxide Research
Ongoing research continues to explore potential new applications of barium oxide. The focus is on developing more sustainable and efficient synthesis methods, improving existing applications, and discovering novel uses in emerging fields like energy storage and advanced materials science.
Conclusion
Barium oxide (BaO), with its simple chemical formula yet diverse properties, plays a significant role in many industries. Understanding its formula, synthesis methods, physical and chemical properties, and safety considerations is crucial for its safe and efficient use. The continued exploration of its properties holds the potential for even more innovative applications in the future. The compound’s versatility and relatively simple chemical makeup make it a subject of ongoing research and development, paving the way for potentially groundbreaking advancements across diverse technological sectors. Remember always to prioritize safety when working with barium oxide due to its inherent reactivity and toxicity.
Latest Posts
Latest Posts
-
Is Sulfur A Metal Metalloid Or Nonmetal
Apr 06, 2025
-
A Group Of Ecosystems With Similar Climates And Organisms
Apr 06, 2025
-
Which Quantum Number Describes The Shape Of An Orbital
Apr 06, 2025
-
Equation Of Line Parallel To Y Axis
Apr 06, 2025
-
Is The Paramecium A Unicellular Or Multicellular Organism
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Enter The Formula For The Compound Barium Oxide. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
