Does Reactivity Increase Down A Group
News Leon
Apr 02, 2025 · 6 min read
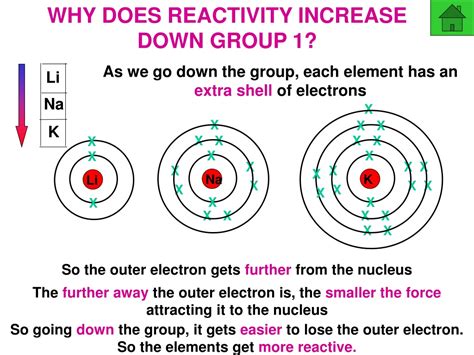
Table of Contents
Does Reactivity Increase Down a Group? Exploring Periodic Trends
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. One of the most fundamental trends observed is the change in reactivity as you move down a group (vertical column). While the general rule states that reactivity increases down a group for metals and decreases for nonmetals, the nuances are far more intricate and depend heavily on the specific group and the type of reaction being considered. This article delves into the complexities of this trend, examining the underlying causes and providing specific examples to illustrate the concept.
Understanding Reactivity
Before exploring the group-wise trend, let's define reactivity. In chemistry, reactivity refers to the tendency of an element or compound to undergo a chemical reaction. A highly reactive substance readily participates in chemical changes, while a less reactive substance is slower or less likely to react. Reactivity is influenced by several factors, including:
- Electron Configuration: The arrangement of electrons in an atom's shells determines its ability to gain, lose, or share electrons, which is the foundation of chemical bonding.
- Ionization Energy: The energy required to remove an electron from an atom. Lower ionization energy generally indicates higher reactivity for metals.
- Electron Affinity: The energy change when an atom gains an electron. Higher electron affinity generally indicates higher reactivity for nonmetals.
- Electronegativity: A measure of an atom's ability to attract electrons in a chemical bond. This is crucial in understanding the reactivity of elements in covalent bonding.
- Atomic Radius: The size of an atom. Larger atomic radius often correlates with increased reactivity for metals due to the shielding effect.
Reactivity of Metals Down a Group: A General Increase
For metals, the general trend is an increase in reactivity down a group. This is primarily attributed to the increasing atomic radius and decreasing ionization energy.
The Role of Atomic Radius and Shielding Effect
As you move down a group, the number of electron shells increases. This leads to a larger atomic radius. The outermost electrons are further from the positively charged nucleus, experiencing a weaker electrostatic attraction. This is further enhanced by the shielding effect, where inner electrons partially shield the outermost electrons from the nuclear charge. Consequently, it becomes easier to remove an electron from the outermost shell, leading to lower ionization energy and increased reactivity.
Ionization Energy and Reactivity
Ionization energy is a crucial factor in determining a metal's reactivity. Metals tend to lose electrons to achieve a stable electron configuration (often a full outer shell). The lower the ionization energy, the easier it is for a metal atom to lose an electron, thus increasing its reactivity. Since ionization energy decreases down a group, reactivity increases.
Examples: Alkali Metals (Group 1) and Alkaline Earth Metals (Group 2)
The alkali metals (Li, Na, K, Rb, Cs, Fr) exemplify this trend perfectly. Lithium is less reactive than sodium, which is less reactive than potassium, and so on. This is evident in their reactions with water; lithium reacts vigorously, sodium more violently, and potassium even more dramatically. A similar trend is observed in the alkaline earth metals (Be, Mg, Ca, Sr, Ba, Ra), although their reactivity is generally lower than that of the alkali metals.
Reactivity of Nonmetals Down a Group: A General Decrease
In contrast to metals, the reactivity of nonmetals generally decreases down a group. This is mainly due to the increasing atomic radius and decreasing electron affinity.
Electron Affinity and Reactivity
Nonmetals tend to gain electrons to achieve a stable electron configuration. Electron affinity measures the energy change associated with gaining an electron. A higher electron affinity indicates a greater tendency to gain an electron, leading to higher reactivity. However, as you move down a group, the atomic radius increases, and the added electron is further from the nucleus, experiencing a weaker attraction. This results in a decrease in electron affinity and therefore a decrease in reactivity.
Examples: Halogens (Group 17) and Chalcogens (Group 16)
The halogens (F, Cl, Br, I, At) clearly demonstrate this trend. Fluorine is the most reactive halogen, followed by chlorine, bromine, iodine, and astatine. Fluorine readily reacts with most other elements, while astatine's reactivity is significantly lower. Similarly, the chalcogens (O, S, Se, Te, Po) show a decrease in reactivity down the group, with oxygen being the most reactive.
Exceptions and Nuances
While the general trends are helpful, it's crucial to acknowledge exceptions and nuances. The reactivity of an element is not solely determined by its position in the periodic table. Other factors, such as:
- Specific reaction conditions: Temperature, pressure, and the presence of catalysts can significantly influence reaction rates.
- The nature of the reacting species: The reactivity of an element can vary depending on the other substance it is reacting with.
- Oxidation states: An element can exhibit multiple oxidation states, and its reactivity can differ depending on the oxidation state.
- d-block and f-block elements: The reactivity of transition metals and inner transition metals is more complex and doesn't always follow the simple trends observed in main group elements.
Analyzing Specific Groups in Detail
Let's explore the reactivity trends in some specific groups:
Group 1: Alkali Metals
The reactivity of alkali metals increases dramatically down the group due to the factors discussed earlier. Their low ionization energies allow them to readily lose their single valence electron, forming +1 ions. This makes them highly reactive with water, oxygen, and halogens.
Group 2: Alkaline Earth Metals
Similar to alkali metals, alkaline earth metals show an increase in reactivity down the group, although less pronounced. They lose two valence electrons to form +2 ions. Their reactivity is lower than alkali metals due to their higher ionization energies.
Group 17: Halogens
Halogens exhibit a decrease in reactivity down the group. Their high electron affinities allow them to readily gain an electron to form -1 ions. Fluorine, with its small size and high electronegativity, is exceptionally reactive.
Group 18: Noble Gases
Noble gases are famously unreactive due to their full valence electron shells. Their extremely high ionization energies prevent them from readily losing electrons. However, even noble gases can participate in reactions under specific conditions.
Conclusion: A Complex Trend
The statement that reactivity increases down a group is a useful generalization, but it's crucial to understand the underlying factors and exceptions. While atomic radius, ionization energy, and electron affinity play significant roles, the specific reaction conditions and the nature of the reacting species can significantly impact the observed reactivity. Therefore, while the periodic trends provide a valuable framework for predicting reactivity, a deeper understanding of the chemical principles involved is necessary for accurate predictions. The complexities inherent in chemical reactions make it essential to approach generalizations with a nuanced perspective, considering the various factors at play.
Latest Posts
Latest Posts
-
Which Of The Following Is An Implicit Cost Of Production
Apr 03, 2025
-
Select The Sentence That Is Punctuated Correctly
Apr 03, 2025
-
Which Of The Following Is The Correct Accounting Equation
Apr 03, 2025
-
Check If Number Is Negative Python
Apr 03, 2025
-
What Is The Measure Of Angle Cbd
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Does Reactivity Increase Down A Group . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
