Ba No3 2 Na2so4 Balanced Equation
News Leon
Apr 02, 2025 · 6 min read
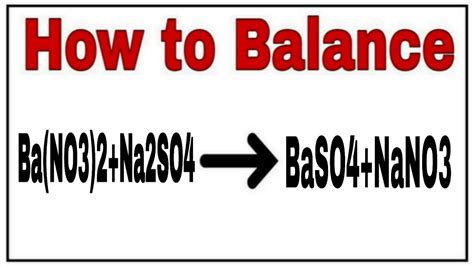
Table of Contents
Ba(NO₃)₂ + Na₂SO₄: A Deep Dive into the Balanced Equation and its Implications
The reaction between barium nitrate (Ba(NO₃)₂) and sodium sulfate (Na₂SO₄) is a classic example of a double displacement reaction, also known as a metathesis reaction. Understanding this reaction, including balancing its equation and exploring its implications, is crucial for students of chemistry and anyone interested in the fundamentals of chemical reactions. This article will provide a comprehensive analysis of the Ba(NO₃)₂ + Na₂SO₄ reaction, covering its balanced equation, the type of reaction, the products formed, and the practical applications and implications of this seemingly simple chemical process.
Understanding the Reactants: Ba(NO₃)₂ and Na₂SO₄
Before delving into the reaction itself, let's briefly examine the individual reactants:
Barium Nitrate (Ba(NO₃)₂)
Barium nitrate is a white crystalline solid that is readily soluble in water. It's an ionic compound consisting of barium cations (Ba²⁺) and nitrate anions (NO₃⁻). Barium compounds are known for their toxicity, and thus handling barium nitrate requires caution. Its primary uses include the production of green fireworks due to the characteristic green flame produced when it's heated. It also finds applications in pyrotechnics and some industrial processes.
Sodium Sulfate (Na₂SO₄)
Sodium sulfate, also known as Glauber's salt (when hydrated), is another white crystalline solid that is highly soluble in water. It is also an ionic compound, composed of sodium cations (Na⁺) and sulfate anions (SO₄²⁻). Unlike barium nitrate, sodium sulfate is relatively non-toxic. It has various industrial applications, including in the paper and textile industries, as well as a laxative in medicine (when hydrated).
The Balanced Chemical Equation
The reaction between barium nitrate and sodium sulfate results in a double displacement reaction, where the cations and anions of the reactants switch partners to form new compounds. The unbalanced equation is:
Ba(NO₃)₂(aq) + Na₂SO₄(aq) → BaSO₄ + NaNO₃
This equation is unbalanced because the number of atoms of each element is not equal on both sides of the arrow. To balance it, we need to ensure that the number of each type of atom is the same on both the reactant and product sides. The balanced equation is:
Ba(NO₃)₂(aq) + Na₂SO₄(aq) → BaSO₄(s) + 2NaNO₃(aq)
This balanced equation shows that one mole of barium nitrate reacts with one mole of sodium sulfate to produce one mole of barium sulfate and two moles of sodium nitrate. The (aq) indicates that the substance is dissolved in water (aqueous solution), while (s) denotes a solid precipitate.
The Products: BaSO₄ and NaNO₃
The products of this reaction are barium sulfate (BaSO₄) and sodium nitrate (NaNO₃). Let's examine each product individually:
Barium Sulfate (BaSO₄)
Barium sulfate is a white, insoluble solid that precipitates out of the solution. This insolubility is a key characteristic of this reaction and is what makes it readily observable. The formation of this precipitate is the driving force behind the reaction. While barium compounds are generally toxic, barium sulfate is an exception due to its very low solubility. This low solubility means that very little barium ions are available for absorption into the body, making it relatively safe. It's commonly used as a contrast agent in medical imaging, particularly in X-rays of the gastrointestinal tract.
Sodium Nitrate (NaNO₃)
Sodium nitrate is a white crystalline solid that remains dissolved in the solution after the reaction. It is highly soluble in water and is a relatively non-toxic compound. Sodium nitrate has various applications, including in fertilizers, as a food preservative, and in some industrial processes.
The Type of Reaction: Double Displacement
As mentioned earlier, this reaction is classified as a double displacement reaction (or metathesis reaction). In this type of reaction, the positive ions (cations) of two different ionic compounds switch places, forming two new ionic compounds. The general form of a double displacement reaction is:
AB + CD → AD + CB
where A and C are cations, and B and D are anions. The reaction between barium nitrate and sodium sulfate perfectly fits this pattern. The driving force for this reaction is the formation of the insoluble barium sulfate precipitate.
Practical Applications and Implications
The reaction between barium nitrate and sodium sulfate, while seemingly simple, has several practical applications and implications:
-
Qualitative Analysis: This reaction is often used in qualitative analysis to identify the presence of either barium ions (Ba²⁺) or sulfate ions (SO₄²⁻). The formation of the white barium sulfate precipitate serves as a positive test for either ion, provided the other ion is known to be present.
-
Quantitative Analysis: The reaction can also be used in quantitative analysis, employing techniques like gravimetric analysis. By carefully measuring the mass of the precipitated barium sulfate, one can determine the concentration of either barium or sulfate ions in a solution.
-
Wastewater Treatment: The precipitation of barium sulfate can potentially be utilized in wastewater treatment to remove sulfate ions. However, the toxicity of barium compounds generally limits its widespread use in this context. More environmentally friendly alternatives are usually preferred.
-
Synthesis of Barium Sulfate: This reaction provides a straightforward method for synthesizing barium sulfate in a laboratory setting. The purity of the product, however, depends on the purity of the starting materials and the careful execution of the procedure.
Further Considerations and Related Reactions
While we have focused primarily on the reaction between barium nitrate and sodium sulfate, it's important to note some related considerations:
-
Other Barium Salts: Similar reactions occur when other soluble barium salts react with soluble sulfate salts. The precipitate formed will always be barium sulfate.
-
Solubility Rules: Understanding solubility rules is crucial for predicting whether a double displacement reaction will occur and whether a precipitate will form. This reaction highlights the importance of learning these rules for predicting the outcome of chemical reactions.
-
Ionic Equations: Writing the net ionic equation for this reaction further clarifies the essential components involved. The net ionic equation eliminates spectator ions (ions that do not participate directly in the reaction), leaving only the ions that are directly involved in the precipitation reaction. The net ionic equation is:
Ba²⁺(aq) + SO₄²⁻(aq) → BaSO₄(s)
This equation clearly shows that the barium and sulfate ions combine to form the insoluble barium sulfate precipitate.
Conclusion: A Foundational Reaction in Chemistry
The reaction between barium nitrate and sodium sulfate, resulting in the formation of barium sulfate and sodium nitrate, serves as a fundamental example of a double displacement reaction. Understanding this reaction, its balanced equation, and the properties of its reactants and products is essential for grasping core concepts in chemistry. From its applications in qualitative and quantitative analysis to its implications in various industrial processes and even medical imaging, this seemingly simple reaction holds significant importance in the broader context of chemical science and technology. The insights gained from studying this reaction can be extended to understanding a wide range of other double displacement reactions and the principles governing the formation of precipitates.
Latest Posts
Latest Posts
-
How Many Protons Neutrons And Electrons In Iron
Apr 03, 2025
-
Which Rule For Assigning Oxidation Numbers Is Correct
Apr 03, 2025
-
Which Of These Characteristics First Developed In Reptiles
Apr 03, 2025
-
Do Annelids Have An Open Or Closed Circulatory System
Apr 03, 2025
-
Which Is Not A Component Of Emotional Intelligence
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Ba No3 2 Na2so4 Balanced Equation . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
