Attraction Among Molecules Of The Same Type Is Called
News Leon
Apr 04, 2025 · 7 min read
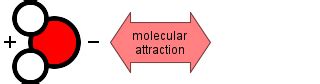
Table of Contents
Attraction Among Molecules of the Same Type is Called Cohesion: A Deep Dive into Intermolecular Forces
Attraction among molecules of the same type is called cohesion. This fundamental concept in chemistry and physics underpins many properties of matter, from the surface tension of water to the strength of materials. Understanding cohesion requires exploring the various intermolecular forces responsible for this attraction, their strengths, and their impact on macroscopic properties. This article will delve into the intricacies of cohesion, examining its underlying mechanisms and diverse manifestations in the natural world.
What is Cohesion?
Cohesion refers to the attractive force between like molecules. These molecules can be of the same substance, like water molecules attracting other water molecules, or similar molecules within a mixture. This attraction arises from the interplay of intermolecular forces, which are weaker than the intramolecular forces (bonds) that hold atoms together within a molecule. However, the cumulative effect of many cohesive forces can be significant, influencing the physical state and behavior of substances. Imagine a drop of water clinging to a surface – this is cohesion in action. The water molecules are sticking together, forming a cohesive droplet.
Intermolecular Forces: The Driving Force Behind Cohesion
Several types of intermolecular forces contribute to cohesion. The strength of these forces dictates the degree of cohesion exhibited by a substance. These forces include:
1. Hydrogen Bonding: A Strong Cohesive Force
Hydrogen bonding is a particularly strong type of dipole-dipole interaction. It occurs when a hydrogen atom bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) is attracted to another electronegative atom in a nearby molecule. Water (H₂O) is a prime example. The hydrogen atom in one water molecule is attracted to the oxygen atom in another, forming a strong cohesive network. This strong cohesion is responsible for many of water's unique properties, including its high surface tension, high boiling point, and ability to act as a universal solvent.
Strong hydrogen bonds lead to high cohesion, influencing properties like:
- High boiling point: More energy is required to overcome the strong cohesive forces and transition from liquid to gas.
- High surface tension: The strong inward pull of the molecules creates a surface "skin".
- Capillary action: Water molecules' ability to climb narrow tubes against gravity.
2. Dipole-Dipole Interactions: Polar Molecule Attraction
Dipole-dipole interactions occur between polar molecules – molecules with a permanent dipole moment due to an uneven distribution of charge. The positive end of one polar molecule is attracted to the negative end of another. While weaker than hydrogen bonds, dipole-dipole interactions still contribute significantly to cohesion in many substances. For instance, acetone (CH₃COCH₃) exhibits significant cohesion due to its polar carbonyl group.
Factors influencing dipole-dipole interactions include:
- Molecular polarity: The larger the dipole moment, the stronger the interaction.
- Molecular shape: Molecular geometry influences the proximity and alignment of dipoles.
3. London Dispersion Forces (LDFs): Universal Cohesive Force
London Dispersion Forces (LDFs), also known as van der Waals forces, are the weakest type of intermolecular force but are present in all molecules, regardless of polarity. They arise from temporary fluctuations in electron distribution, creating temporary dipoles. These temporary dipoles induce dipoles in neighboring molecules, leading to a weak attractive force. While individually weak, the cumulative effect of LDFs can be substantial, especially in large molecules with many electrons.
LDF strength depends on:
- Molecular size and shape: Larger molecules with more electrons have stronger LDFs.
- Molecular surface area: A larger surface area allows for more contact and interaction between molecules.
4. Ion-Dipole Interactions: Cohesion Involving Ions
When ions are present in a polar solvent, ion-dipole interactions contribute to cohesion. The positive or negative ions are attracted to the oppositely charged ends of the polar molecules. This is crucial in solutions involving ionic compounds dissolved in polar solvents like water. The strong attraction between ions and water molecules helps dissolve the ionic compound and contributes to the overall cohesion of the solution.
Manifestations of Cohesion in Everyday Life
Cohesion is not just a theoretical concept; it plays a crucial role in numerous everyday phenomena:
- Surface tension of water: Water's high surface tension, due to strong hydrogen bonding, allows insects to walk on water and enables the formation of water droplets.
- Capillary action: The ability of water to rise in narrow tubes, like plant stems, is driven by cohesion and adhesion (attraction to other substances).
- Rain droplets: Cohesion holds water molecules together, forming spherical droplets that minimize surface area.
- The strength of materials: Cohesion contributes to the overall strength and integrity of solid materials. Strong cohesive forces within a solid provide structural integrity and resistance to deformation.
- Viscosity of liquids: The resistance of a liquid to flow is influenced by cohesive forces. Liquids with strong cohesive forces tend to have higher viscosity.
- Boiling point and melting point: The temperature at which a substance changes its phase (solid to liquid or liquid to gas) depends largely on the strength of its cohesive forces. Substances with stronger cohesive forces require more energy to overcome these forces and undergo phase transitions.
Cohesion vs. Adhesion: A Key Distinction
It's important to distinguish between cohesion and adhesion, which is the attractive force between different molecules. While cohesion involves the attraction between like molecules, adhesion involves the attraction between unlike molecules. Both forces are essential in many phenomena. For example, capillary action is a result of the interplay between cohesion and adhesion: water molecules stick to each other (cohesion) and to the walls of the tube (adhesion), allowing the water to climb upwards.
Measuring Cohesion: Techniques and Applications
Measuring the precise strength of cohesive forces is complex and depends on the type of intermolecular force dominant in the substance. Several techniques can provide insight into cohesive strength:
- Surface tension measurements: Techniques like the du Noüy ring method or Wilhelmy plate method measure the force required to break the surface of a liquid, reflecting its cohesive strength.
- Viscosity measurements: Measuring the viscosity of a liquid provides information about the strength of cohesive forces resisting flow.
- Boiling point determination: The boiling point directly correlates with the strength of cohesive forces, providing a simple yet useful indirect measure.
- Computational methods: Molecular dynamics simulations and quantum chemical calculations can predict and analyze intermolecular forces and their contribution to cohesion.
These techniques find applications in various fields, including materials science, chemical engineering, and environmental science. For example, understanding the cohesion of polymers is crucial in designing strong and durable materials, while assessing the cohesion of soil particles is essential in agriculture and environmental management.
The Importance of Cohesion in Biology
Cohesion plays a vital role in various biological processes:
- Water transport in plants: Cohesion and adhesion are crucial for the upward movement of water in plants through xylem vessels.
- Cell structure and function: Cohesion forces maintain the structural integrity of cells and tissues.
- Protein folding: Cohesion contributes to the folding and stability of proteins, essential for their function.
- DNA structure: Hydrogen bonding, a type of cohesive force, plays a crucial role in the double helix structure of DNA.
Understanding cohesion at a molecular level is crucial in various biological research fields, including biochemistry, molecular biology, and cell biology.
Conclusion: Cohesion—A Fundamental Force Shaping Our World
Cohesion, the attractive force between like molecules, is a fundamental force shaping the world around us. Its impact ranges from the microscopic level, influencing the structure and function of biological molecules, to the macroscopic level, determining the properties of materials and influencing natural phenomena. Understanding the various intermolecular forces that contribute to cohesion is essential to comprehending the behavior of matter and its role in biological systems and technological applications. Further research into cohesive forces continues to reveal its significance in various scientific domains, promising exciting advancements in materials science, nanotechnology, and biotechnology.
Latest Posts
Latest Posts
-
8 Scientists Who Contributed To The Atomic Theory
Apr 04, 2025
-
How Do Strict Constructionists And Liberal Constructionists Differ
Apr 04, 2025
-
Breakdown Of Glucose To Pyruvic Acid
Apr 04, 2025
-
Are The Following Two Figures Similar Or Congruent
Apr 04, 2025
-
Python Round Number To 2 Decimals
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Attraction Among Molecules Of The Same Type Is Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
