An Electrically Charged Atom Or Group Of Atoms
News Leon
Apr 05, 2025 · 7 min read
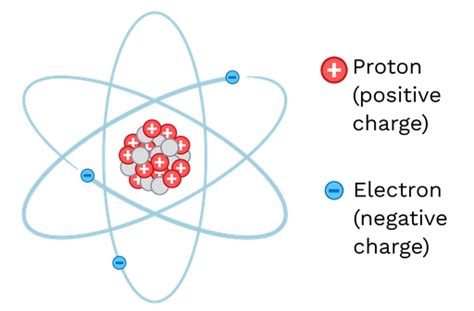
Table of Contents
An Electrically Charged Atom or Group of Atoms: Exploring Ions and Their Significance
An electrically charged atom or group of atoms is known as an ion. Ions are fundamental building blocks of matter, playing crucial roles in numerous chemical and biological processes. Understanding their properties, formation, and behavior is vital across diverse scientific fields, from chemistry and physics to biology and materials science. This article delves deep into the world of ions, exploring their characteristics, types, formation mechanisms, and significant applications.
What are Ions?
At their core, ions are atoms or molecules that carry a net electrical charge. This charge arises from an imbalance in the number of protons (positively charged particles) and electrons (negatively charged particles) within the atom or molecule. A neutral atom possesses an equal number of protons and electrons, resulting in a net charge of zero. However, when an atom gains or loses electrons, it becomes an ion, acquiring a positive or negative charge, respectively.
Cations and Anions: Two Sides of the Ion Coin
Ions are broadly classified into two categories based on their charge:
-
Cations: These are positively charged ions, formed when an atom loses one or more electrons. The loss of negatively charged electrons leaves the atom with a surplus of positive charge from the protons. Metals, with their relatively low electronegativity, readily form cations. Examples include Na⁺ (sodium ion), Ca²⁺ (calcium ion), and Fe³⁺ (iron(III) ion).
-
Anions: These are negatively charged ions, formed when an atom gains one or more electrons. The addition of negatively charged electrons results in an excess of negative charge. Non-metals, with their higher electronegativity, tend to form anions. Examples include Cl⁻ (chloride ion), O²⁻ (oxide ion), and SO₄²⁻ (sulfate ion).
The Formation of Ions: A Matter of Electron Transfer
The formation of ions is a process governed by the principles of electron transfer. This transfer can occur through various mechanisms, including:
-
Electron Transfer in Chemical Reactions: Many chemical reactions involve the transfer of electrons between atoms or molecules. For instance, in the reaction between sodium (Na) and chlorine (Cl), sodium readily loses an electron to become a sodium cation (Na⁺), while chlorine gains an electron to become a chloride anion (Cl⁻). This transfer leads to the formation of an ionic compound, sodium chloride (NaCl), also known as table salt.
-
Ionization by Radiation: Exposure to high-energy radiation, such as X-rays or gamma rays, can ionize atoms. The radiation's energy can knock electrons out of atoms, leaving behind positively charged ions. This process is fundamental to various techniques like X-ray fluorescence spectroscopy and ionization detectors in radiation monitoring.
-
Thermal Ionization: At extremely high temperatures, the kinetic energy of atoms becomes sufficient to overcome the electrostatic attraction between electrons and the nucleus. This leads to the ejection of electrons, forming ions. This is prevalent in plasmas, high-temperature environments like stars, and in certain industrial processes.
Properties of Ions
Ions possess distinct properties that differ from their neutral counterparts:
-
Electrical Charge: The most defining characteristic of an ion is its net electrical charge, which dictates its interactions with electric and magnetic fields. Cations are attracted to negatively charged electrodes (cathodes), while anions are attracted to positively charged electrodes (anodes).
-
Size: The size of an ion differs from that of its neutral atom. Cations are generally smaller than their parent atoms because they have lost electrons, reducing electron-electron repulsion and allowing the remaining electrons to be pulled closer to the nucleus. Conversely, anions are generally larger than their parent atoms due to the added electrons, increasing electron-electron repulsion and expanding the electron cloud.
-
Reactivity: Ions exhibit high reactivity, readily participating in chemical reactions. Their charge allows them to form ionic bonds with oppositely charged ions, creating ionic compounds. The strength of these bonds depends on the charges and sizes of the ions involved.
-
Solubility: The solubility of ionic compounds varies depending on the nature of the ions and the solvent. Many ionic compounds are soluble in polar solvents like water, where the polar water molecules can effectively interact with and stabilize the charged ions.
Significance of Ions in Various Fields
Ions play crucial roles in a vast array of fields:
Biology and Medicine
-
Electrolyte Balance: Ions like sodium (Na⁺), potassium (K⁺), calcium (Ca²⁺), and chloride (Cl⁻) are essential electrolytes in biological systems. They maintain fluid balance, regulate nerve impulses, muscle contractions, and enzyme activity. Electrolyte imbalances can lead to serious health problems.
-
Cellular Processes: Many biological processes, including nerve transmission, muscle contraction, and nutrient transport across cell membranes, rely on the movement of ions across cell membranes. These movements are facilitated by ion channels and pumps, which regulate ion concentrations within and outside cells.
-
Medical Imaging: Techniques like magnetic resonance imaging (MRI) utilize the magnetic properties of ions to generate detailed images of the human body.
Chemistry and Materials Science
-
Ionic Compounds: Ions are the fundamental building blocks of ionic compounds, a vast class of materials with diverse properties and applications. These compounds range from table salt (NaCl) to complex minerals and ceramics.
-
Electrochemistry: Electrochemistry utilizes the movement of ions to generate electricity (batteries) and carry out chemical reactions (electrolysis). Ions are central to the operation of batteries, fuel cells, and various electrochemical sensors.
-
Materials Synthesis: The controlled introduction of specific ions into materials during synthesis can drastically alter their properties. For instance, doping semiconductors with specific ions can enhance their conductivity or optical properties.
Environmental Science
-
Water Quality: The presence and concentration of specific ions in water determine its quality and suitability for various purposes. Testing for ions like nitrates, phosphates, and heavy metal ions is crucial for assessing water pollution and ensuring safe drinking water.
-
Atmospheric Chemistry: Ions play an essential role in atmospheric processes, influencing cloud formation, precipitation, and air pollution. Understanding ion chemistry in the atmosphere is vital for modelling climate change and air quality.
Polyatomic Ions: The Complex Family
Beyond simple monoatomic ions, there exists a vast array of polyatomic ions. These are ions composed of multiple atoms covalently bonded together and carrying a net electrical charge. Examples include:
- Hydroxide ion (OH⁻): Found in bases and involved in many chemical reactions.
- Sulfate ion (SO₄²⁻): A common anion in many minerals and salts.
- Nitrate ion (NO₃⁻): Essential nutrient for plant growth.
- Phosphate ion (PO₄³⁻): Crucial component of DNA and ATP, the cell's energy currency.
- Ammonium ion (NH₄⁺): A common cation found in fertilizers and many organic compounds.
Polyatomic ions exhibit properties similar to monoatomic ions, including electrical charge, reactivity, and solubility. However, their complex structures and internal bonding influence their behavior in unique ways. Understanding the behavior of polyatomic ions is critical in many fields, including biochemistry, environmental chemistry, and materials science.
Analyzing and Detecting Ions: Techniques and Methods
Several analytical techniques are employed to detect and quantify ions in various samples:
-
Flame Emission Spectroscopy: This technique measures the characteristic wavelengths of light emitted by excited ions in a flame.
-
Atomic Absorption Spectroscopy: This method measures the absorption of light by ions in a gaseous sample.
-
Ion Chromatography: This technique separates and quantifies ions based on their interaction with a stationary phase.
-
Mass Spectrometry: This powerful technique separates ions based on their mass-to-charge ratio, providing detailed information about the composition and structure of ionic species.
These and other techniques allow scientists to accurately determine the types and amounts of ions present in a wide variety of samples, from biological fluids to environmental samples to industrial materials.
Conclusion: Ions - The Unsung Heroes of Chemistry and Beyond
Electrically charged atoms and groups of atoms, or ions, are far more than mere abstractions in chemistry textbooks. They are fundamental components of matter, driving a wide array of processes that shape our world and sustain life itself. Their importance spans numerous fields, from the intricate mechanisms of life to the design of advanced materials and the understanding of environmental challenges. Continued research into the properties and behavior of ions will undoubtedly unveil even more fascinating insights into the natural world and pave the way for innovative applications in science and technology. Understanding ions is key to understanding the universe around us and harnessing its power for the benefit of humanity.
Latest Posts
Latest Posts
-
How Does Ka Relate To Acid Strength
Apr 06, 2025
-
Which Molecule Has A Higher Potential Energy
Apr 06, 2025
-
Is Energy Recycled In The Ecosystem
Apr 06, 2025
-
How To Separate Sand From Sugar
Apr 06, 2025
-
Cannot Be Separated By Physical Means
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about An Electrically Charged Atom Or Group Of Atoms . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
