A Red Blood Cell Placed In A Hypotonic Solution
News Leon
Apr 04, 2025 · 6 min read
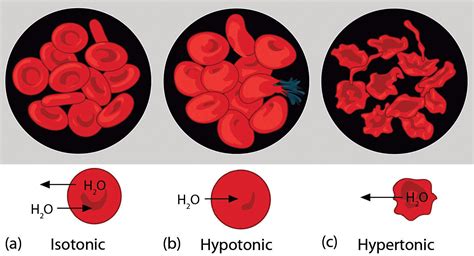
Table of Contents
A Red Blood Cell Placed in a Hypotonic Solution: A Deep Dive into Osmosis and Hemolysis
Red blood cells, the tireless workhorses of our circulatory system, are fascinating biological entities. Their structure and function are intimately tied to the surrounding environment, particularly the osmotic pressure of the solution they're immersed in. Understanding how a red blood cell (RBC), also known as an erythrocyte, behaves in different solutions is crucial to grasping fundamental concepts in biology and medicine. This article delves deep into the consequences of placing a red blood cell in a hypotonic solution, exploring the underlying mechanisms and broader implications.
Understanding Osmosis and Tonicity
Before we dissect the effects of a hypotonic solution on an RBC, let's establish a strong foundation in osmosis and tonicity. Osmosis is the passive movement of water across a selectively permeable membrane from a region of high water concentration (low solute concentration) to a region of low water concentration (high solute concentration). This movement continues until equilibrium is reached, meaning the water concentration is equal on both sides of the membrane.
Tonicity refers to the relative concentration of solutes in two solutions separated by a selectively permeable membrane. We typically compare the solute concentration of the solution surrounding the cell (the extracellular fluid) to the solute concentration inside the cell (the intracellular fluid). There are three main types of tonicity:
- Hypotonic: The extracellular solution has a lower solute concentration than the intracellular solution. This means the water concentration is higher outside the cell.
- Isotonic: The extracellular solution has the same solute concentration as the intracellular solution. Water movement is equal in both directions.
- Hypertonic: The extracellular solution has a higher solute concentration than the intracellular solution. This means the water concentration is higher inside the cell.
What Happens When a Red Blood Cell is Placed in a Hypotonic Solution?
When a red blood cell is placed in a hypotonic solution, the concentration of water is higher outside the cell than inside. Due to osmosis, water rushes into the RBC across its cell membrane. The cell membrane, while selectively permeable, allows water to pass relatively freely.
The Initial Stages: Water Influx and Cell Swelling
The initial response is a gradual increase in the cell's volume as it takes in water. The cell membrane, being flexible, stretches to accommodate the increasing volume. This swelling is not immediately catastrophic. The cell's internal pressure (turgor pressure) rises, counteracting the osmotic pressure pulling water in.
The Point of No Return: Hemolysis
However, the cell's membrane has its limits. If the hypotonic solution is significantly less concentrated than the cell's cytoplasm, the influx of water overwhelms the cell's capacity to withstand the increased internal pressure. This leads to hemolysis, the rupture of the red blood cell. The cell's membrane breaks, releasing hemoglobin and other intracellular contents into the surrounding solution. The result is a clear solution tinged with a reddish hue.
Factors Influencing Hemolysis Rate
The rate at which hemolysis occurs depends on several factors:
- The degree of hypotonic nature of the solution: The greater the difference in solute concentration between the solution and the cell's interior, the faster the hemolysis.
- The duration of exposure: Prolonged exposure to a hypotonic solution increases the likelihood of hemolysis.
- The cell's age and health: Older or damaged RBCs may be more susceptible to hemolysis.
- The presence of other substances: Certain substances can affect the cell membrane's integrity, making it more or less prone to rupture.
Microscopic Observation of Hemolysis
Observing hemolysis under a microscope provides a visual confirmation of this process. Initially, you would observe normal, biconcave RBCs. As water enters the cell, they swell and become more spherical. Finally, the membrane ruptures, and the cell's contents are dispersed, leaving behind only "ghosts" or empty cell membranes.
Physiological Significance and Medical Implications
Understanding hemolysis in hypotonic solutions has significant implications in various physiological and medical contexts.
Maintaining Blood Osmotic Pressure
The human body maintains a delicate balance of osmotic pressure in the blood. Deviations from this balance, such as exposure of red blood cells to hypotonic conditions, can have serious consequences. Maintaining isotonic conditions is crucial for normal RBC function and survival.
Intravenous Fluid Administration
In clinical settings, the correct administration of intravenous (IV) fluids is paramount. Administering hypotonic solutions intravenously could lead to hemolysis in the bloodstream, causing potentially life-threatening complications. Healthcare professionals meticulously choose isotonic solutions to avoid this risk.
Blood Transfusions
During blood transfusions, careful matching of blood types is essential to prevent agglutination (clumping) and subsequent hemolysis. If incompatible blood types are mixed, the recipient's immune system may attack the transfused cells, leading to hemolysis and potentially fatal complications.
Water Intoxication (Hyponatremia)
Excessive water intake without a corresponding increase in electrolytes can dilute the blood, creating a hypotonic environment. This condition, known as hyponatremia or water intoxication, can lead to hemolysis and other serious health consequences. Symptoms can range from nausea and headaches to seizures and coma.
Kidney Disease
Kidney dysfunction can impact the body's ability to regulate electrolyte balance, potentially leading to hypotonic conditions and hemolysis. Impaired kidney function prevents effective regulation of water and solute levels in the blood, thereby increasing the risk of hemolysis.
Genetic Disorders
Certain genetic disorders affect the structure and function of the red blood cell membrane, rendering them more vulnerable to hemolysis in hypotonic or even isotonic environments. These disorders often manifest as various types of hemolytic anemia.
Experimental Demonstrations and Applications
Hemolysis in hypotonic solutions is frequently demonstrated in introductory biology and physiology labs. Students can directly observe the process by placing RBCs in solutions of varying tonicity and observing the changes under a microscope.
These experiments help students visualize osmotic pressure, cell membrane function, and the importance of maintaining fluid balance. They provide a hands-on understanding of fundamental biological principles and their relevance to human health.
Conclusion: The Importance of Osmotic Balance
The behavior of a red blood cell in a hypotonic solution vividly illustrates the importance of osmotic balance for cellular health and overall physiological function. The delicate equilibrium between the intracellular and extracellular environments is crucial for maintaining the integrity of red blood cells and their essential role in oxygen transport. A thorough understanding of osmosis and tonicity is therefore vital not only in biology but also in various aspects of medicine, emphasizing the critical role of maintaining isotonic conditions to prevent hemolysis and its potentially serious consequences. Further research continues to explore the nuances of osmotic regulation and the mechanisms that protect cells from osmotic stress, revealing more insights into the intricate workings of biological systems. Continued exploration in this area is essential for developing novel therapeutic strategies and diagnostic tools for diseases related to osmotic imbalances.
Latest Posts
Latest Posts
-
The Function Of The Enzyme Atp Synthase Is To
Apr 05, 2025
-
What Is The Oxidation Number Of Chlorine In Hclo4
Apr 05, 2025
-
A Saprobe Differs From A Parasite In That
Apr 05, 2025
-
What Happens To Plant Cells In A Hypertonic Solution
Apr 05, 2025
-
What Is The Lightest Subatomic Particle
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about A Red Blood Cell Placed In A Hypotonic Solution . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
