A Compound Is 54.53 C 9.15 H
News Leon
Apr 05, 2025 · 5 min read
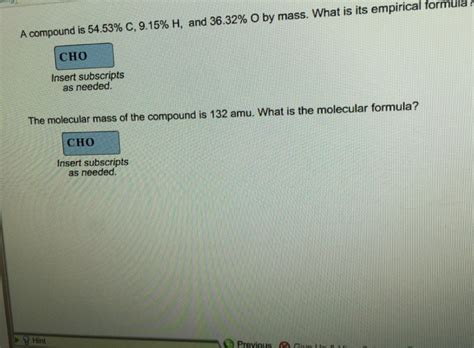
Table of Contents
Unveiling the Mystery: A Compound with 54.53% C and 9.15% H
Determining the identity of an unknown compound is a fundamental task in chemistry. This article delves into the process of identifying a compound with elemental composition of 54.53% Carbon (C) and 9.15% Hydrogen (H). We'll explore the steps involved, from initial analysis to potential compound identification, highlighting the crucial role of chemical principles and analytical techniques. This comprehensive guide aims to provide a clear and detailed understanding of the process, emphasizing practical applications and considerations for accurate results.
Step 1: Calculating the Empirical Formula
The first step in identifying an unknown compound from its elemental composition is to determine its empirical formula. This represents the simplest whole-number ratio of atoms of each element present in the compound. To calculate this, we'll assume a 100g sample of the compound, allowing us to convert percentages directly into grams:
- Carbon (C): 54.53 g
- Hydrogen (H): 9.15 g
Next, we convert these masses into moles using the atomic masses of Carbon (12.01 g/mol) and Hydrogen (1.01 g/mol):
- Moles of C: 54.53 g / 12.01 g/mol = 4.54 moles
- Moles of H: 9.15 g / 1.01 g/mol = 9.06 moles
Now, we find the simplest whole-number ratio by dividing both values by the smaller number (4.54 moles):
- C: 4.54 moles / 4.54 moles = 1
- H: 9.06 moles / 4.54 moles ≈ 2
Therefore, the empirical formula of the compound is CH₂.
Step 2: Determining the Molecular Formula
The empirical formula represents the simplest ratio, but the actual molecular formula may be a multiple of this ratio. To determine the molecular formula, we need additional information, specifically the molar mass (molecular weight) of the compound. This can be obtained experimentally through techniques like mass spectrometry.
Let's assume, for the sake of this example, that the molar mass of the unknown compound is experimentally determined to be 70 g/mol. We can now calculate the empirical formula mass:
- Empirical Formula Mass (CH₂): 12.01 g/mol (C) + 2 * 1.01 g/mol (H) = 14.03 g/mol
Next, we divide the molar mass by the empirical formula mass to find the multiplier:
- Multiplier: 70 g/mol / 14.03 g/mol ≈ 5
Finally, we multiply the subscripts in the empirical formula by this multiplier to obtain the molecular formula:
- Molecular Formula: C₅H₁₀
Step 3: Identifying Possible Compounds
With the molecular formula C₅H₁₀, we can now explore potential compounds that fit this formula. This is where knowledge of organic chemistry and isomerism becomes crucial. C₅H₁₀ represents a class of hydrocarbons known as alkenes (containing a carbon-carbon double bond) and cycloalkanes (containing a ring structure).
Several isomers are possible for C₅H₁₀:
Alkenes:
- 1-Pentene: A straight-chain alkene with the double bond at the terminal carbon.
- 2-Pentene: A straight-chain alkene with the double bond at the second carbon (exists as cis and trans isomers).
- 2-Methyl-1-butene: A branched-chain alkene.
- 3-Methyl-1-butene: A branched-chain alkene.
- 2-Methyl-2-butene: A branched-chain alkene.
Cycloalkanes:
- Cyclopentane: A five-membered ring alkane.
- Methylcyclobutane: A four-membered ring with a methyl group.
- Ethylcyclopropane: A three-membered ring with an ethyl group.
- 1,1-Dimethylcyclopropane: A three-membered ring with two methyl groups.
- cis-1,2-Dimethylcyclopropane: A three-membered ring with two methyl groups on the same side of the ring.
- trans-1,2-Dimethylcyclopropane: A three-membered ring with two methyl groups on opposite sides of the ring.
Step 4: Utilizing Spectroscopic Techniques for Confirmation
To definitively identify the compound, we need to employ spectroscopic techniques such as:
Nuclear Magnetic Resonance (NMR) Spectroscopy:
NMR spectroscopy provides detailed information about the carbon and hydrogen atoms in the molecule, including their chemical environment and connectivity. The NMR spectrum will reveal the number of different types of hydrogens and carbons present, their chemical shifts, and coupling patterns. This data is crucial for distinguishing between the various isomers. For instance, the NMR spectrum of 1-pentene will be distinctly different from that of cyclopentane.
Infrared (IR) Spectroscopy:
IR spectroscopy can identify functional groups present in the molecule. For example, it can differentiate between alkenes (showing C=C stretching) and cycloalkanes (lacking this specific peak).
Mass Spectrometry (MS):
Although we used the mass spectrometry data earlier to determine the molar mass, the fragmentation pattern in the mass spectrum can also be used to distinguish between isomers. Each isomer will produce a unique fragmentation pattern, enabling us to pinpoint the exact structure.
Step 5: Interpreting Results and Reaching a Conclusion
By carefully analyzing the NMR, IR, and MS spectral data, we can pinpoint the exact structural isomer of the C₅H₁₀ compound. The interpretation of these spectra requires expertise and experience in interpreting spectral data, often using software tools for assistance.
For example, a strong peak around 1650 cm⁻¹ in the IR spectrum would indicate the presence of a C=C double bond, pointing towards one of the alkene isomers. The NMR spectrum would then provide further information about the arrangement of atoms around the double bond and the overall molecular structure.
Conclusion:
Identifying an unknown compound based solely on elemental composition requires a multi-step approach combining chemical calculations, spectral analysis, and a thorough understanding of organic chemistry principles. While determining the empirical and molecular formulas provides a starting point, spectroscopic techniques are crucial for confirming the identity of the compound among the many potential isomers. Each step is vital, showcasing the interconnected nature of theoretical and practical aspects of chemistry in solving real-world problems. This comprehensive approach highlights the power of scientific method and the importance of utilizing various analytical techniques in the field of chemical analysis. The combination of these techniques provides a robust and reliable method for identifying unknown compounds, paving the way for advancements in various scientific fields. Further research on the specific properties and applications of the identified compound can also then be undertaken.
Latest Posts
Latest Posts
-
Write Iupac Name Of The Following Compound
Apr 06, 2025
-
Which Of The Following Is Not A Computer Peripheral
Apr 06, 2025
-
The Most Abundant Negative Ion In Blood Plasma Is
Apr 06, 2025
-
What Is The Mass Of An Electron In Amu
Apr 06, 2025
-
What Would Happen To The Cell If Nucleus Is Removed
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about A Compound Is 54.53 C 9.15 H . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
