Which Proportionality Applies To Avogadro's Law
News Leon
Apr 02, 2025 · 5 min read
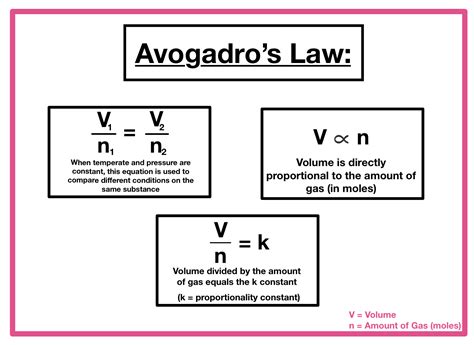
Table of Contents
Which Proportionality Applies to Avogadro's Law? A Deep Dive
Avogadro's Law, a cornerstone of chemistry, describes the relationship between the volume and the amount of gas under specific conditions. Understanding which proportionality governs this relationship is crucial for grasping its implications and applications. This article delves deep into the mathematical representation of Avogadro's Law, exploring its proportionality, underlying assumptions, and limitations, ultimately providing a comprehensive understanding of its applicability.
Understanding Avogadro's Law: A Simple Explanation
Avogadro's Law states that equal volumes of all gases, at the same temperature and pressure, contain the same number of molecules. This seemingly simple statement has profound consequences for our understanding of gases and their behavior. It establishes a direct proportionality between the volume (V) of a gas and the number of moles (n) of the gas, provided temperature (T) and pressure (P) remain constant.
The Mathematical Representation: Direct Proportionality
The mathematical expression of Avogadro's Law clearly illustrates the direct proportionality:
V ∝ n (at constant T and P)
This means that if the number of moles of gas increases, the volume will increase proportionally, and vice versa. If you double the number of moles, keeping temperature and pressure constant, you will double the volume. This relationship is fundamental to many stoichiometric calculations and gas law applications.
Understanding the Constant of Proportionality
The proportionality symbol (∝) can be replaced with an equals sign by introducing a constant of proportionality (k):
V = kn (at constant T and P)
This constant, k, is dependent on the temperature and pressure. While Avogadro's Law focuses on the relationship between volume and moles at constant temperature and pressure, it's important to remember that changing T or P will alter the value of k, impacting the volume-mole relationship.
Ideal Gas Law and Avogadro's Law: A Synergistic Relationship
Avogadro's Law is a special case of the Ideal Gas Law, a more comprehensive equation describing the behavior of ideal gases. The Ideal Gas Law is expressed as:
PV = nRT
Where:
- P = Pressure
- V = Volume
- n = Number of moles
- R = Ideal gas constant
- T = Temperature
If we hold temperature (T) and pressure (P) constant, the Ideal Gas Law simplifies directly to Avogadro's Law:
V = (R*T/P) * n
In this equation, (R*T/P) acts as our constant of proportionality (k). This demonstrates the inherent connection between Avogadro's Law and the more general Ideal Gas Law.
Assumptions and Limitations of Avogadro's Law
It's crucial to acknowledge the assumptions underlying Avogadro's Law:
-
Ideal Gas Behavior: Avogadro's Law assumes that the gas behaves ideally. This means the gas molecules occupy negligible volume compared to the container's volume and that intermolecular forces are negligible. Real gases deviate from ideal behavior, particularly at high pressures and low temperatures.
-
Constant Temperature and Pressure: Maintaining constant temperature and pressure is critical. Any changes in T or P will invalidate the direct proportionality between volume and the number of moles.
-
Homogenous Gas Mixture: Avogadro's Law is applicable to homogenous mixtures of gases. The law assumes all gas molecules in the mixture behave independently.
Deviation from Ideal Behavior: Real Gases
Real gases exhibit intermolecular forces and have non-negligible molecular volumes. At high pressures, the volume occupied by the gas molecules themselves becomes significant, leading to deviations from Avogadro's Law. At low temperatures, intermolecular forces become more prominent, again causing deviations. The van der Waals equation is a more accurate model for describing the behavior of real gases and accounts for these deviations.
Applications of Avogadro's Law: Practical Uses
Avogadro's Law finds numerous applications in various fields:
-
Stoichiometry: It's essential in stoichiometric calculations involving gases. Knowing the volume of one gas allows for the determination of the volume of other gases involved in a reaction, assuming the same temperature and pressure.
-
Gas Analysis: Avogadro's Law plays a crucial role in gas analysis techniques. The volume of a gas can be used to determine its amount, allowing for the composition analysis of gas mixtures.
-
Environmental Monitoring: Monitoring atmospheric gases, like pollutants, often involves applying Avogadro's Law to determine concentrations based on measured volumes.
-
Industrial Processes: Many industrial processes involving gases, such as ammonia synthesis or hydrocarbon processing, rely heavily on Avogadro's Law for efficient control and optimization.
-
Aerospace Engineering: Calculations related to the behavior of gases in aircraft cabins and rocket propulsion systems utilize principles derived from Avogadro's Law.
Beyond the Basics: Advanced Considerations
While Avogadro's Law provides a simple and useful model, it's essential to appreciate its limitations and the complexities of gas behavior. Understanding the relationship between Avogadro's Law and the Ideal Gas Law enables a more nuanced understanding of gas behavior under various conditions.
The Role of the Ideal Gas Constant (R)
The Ideal Gas Constant (R) is crucial in bridging Avogadro's Law and the Ideal Gas Law. Its value depends on the units used for pressure, volume, temperature, and the number of moles. Different values of R are used depending on the units employed in the calculation. The consistency of units is critical for accurate calculations.
Molar Volume: A Direct Consequence of Avogadro's Law
A significant consequence of Avogadro's Law is the concept of molar volume. At standard temperature and pressure (STP), one mole of any ideal gas occupies approximately 22.4 liters. This molar volume serves as a convenient conversion factor between the number of moles and the volume of a gas at STP. Remember that this value is only valid for ideal gases at STP.
Conclusion: A Foundation for Understanding Gases
Avogadro's Law, despite its limitations, provides a fundamental understanding of the relationship between the volume and the amount of gas under constant temperature and pressure. The direct proportionality between volume and the number of moles (at constant T and P) is central to its application. Its connection to the Ideal Gas Law further solidifies its importance in chemistry and related fields. However, always remember that real gases deviate from ideal behavior, especially under extreme conditions. Understanding these limitations ensures accurate applications and interpretations of Avogadro's Law in practical scenarios. This deep understanding allows for accurate predictions and calculations, making Avogadro's Law a powerful tool in various scientific and industrial applications. A thorough grasp of its underlying principles and limitations is essential for anyone studying or working with gases.
Latest Posts
Latest Posts
-
The Price Elasticity Of Demand Is Generally
Apr 03, 2025
-
The Distance Around A Figure Is The
Apr 03, 2025
-
Describe The Role Of Producers In An Ecosystem
Apr 03, 2025
-
What Is The Name For Fecl3
Apr 03, 2025
-
Determine Whether The Following Statement Is True Or False
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Proportionality Applies To Avogadro's Law . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
