Which Of The Following Molecules Has The Highest Boiling Point
News Leon
Apr 05, 2025 · 5 min read
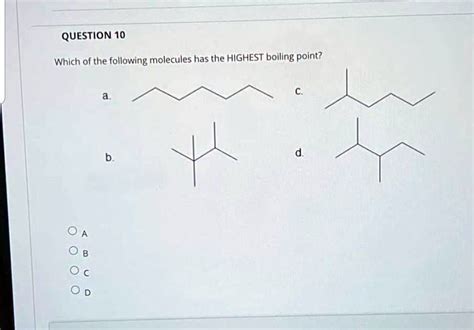
Table of Contents
Which of the Following Molecules Has the Highest Boiling Point? A Deep Dive into Intermolecular Forces
Determining which molecule in a given set boasts the highest boiling point requires a nuanced understanding of intermolecular forces (IMFs). Boiling point, the temperature at which a liquid transitions to a gas, is directly correlated to the strength of these forces. The stronger the IMFs, the more energy (and thus higher temperature) is required to overcome them and initiate boiling. This article will explore the various types of IMFs and delve into how they influence boiling point, ultimately equipping you to confidently answer the question posed in the title for any set of molecules.
Understanding Intermolecular Forces
Before we tackle specific examples, let's review the major types of intermolecular forces:
1. London Dispersion Forces (LDFs): Present in all molecules, LDFs are the weakest type of IMF. They arise from temporary, instantaneous dipoles created by the random movement of electrons within a molecule. These fleeting dipoles induce dipoles in neighboring molecules, leading to a weak attraction. The strength of LDFs increases with the size and shape of the molecule. Larger molecules with greater surface area have more electrons, resulting in stronger, more frequent temporary dipoles. A more elongated shape allows for greater contact between molecules, further strengthening the LDFs.
2. Dipole-Dipole Forces: These forces occur between polar molecules – molecules with a permanent dipole moment due to an uneven distribution of electron density. The positive end of one polar molecule is attracted to the negative end of another. Dipole-dipole forces are stronger than LDFs but weaker than hydrogen bonding. The magnitude of the dipole moment directly impacts the strength of these forces.
3. Hydrogen Bonding: A special type of dipole-dipole interaction, hydrogen bonding is the strongest type of IMF. It occurs when a hydrogen atom bonded to a highly electronegative atom (fluorine, oxygen, or nitrogen) is attracted to a lone pair of electrons on another electronegative atom in a nearby molecule. The high electronegativity of F, O, and N creates a significant partial positive charge on the hydrogen atom, leading to a strong attraction.
Factors Influencing Boiling Point
Several factors influence a molecule's boiling point, all ultimately tied to the strength of its IMFs:
-
Molecular Weight: Larger molecules generally have higher boiling points due to stronger LDFs. The increased number of electrons leads to more frequent and stronger temporary dipoles.
-
Molecular Shape: As mentioned earlier, elongated molecules often have higher boiling points than compact molecules with similar molecular weights due to increased surface area for LDF interactions.
-
Polarity: Polar molecules exhibit dipole-dipole forces in addition to LDFs, leading to higher boiling points compared to nonpolar molecules of similar molecular weight.
-
Hydrogen Bonding: The presence of hydrogen bonding significantly increases boiling points. The strength of hydrogen bonding is considerably greater than other IMFs, requiring more energy to overcome.
Comparing Boiling Points: A Case Study
Let's consider a hypothetical example to illustrate the principles discussed above. Suppose we need to compare the boiling points of the following molecules:
- Methane (CH₄)
- Ethanol (CH₃CH₂OH)
- Water (H₂O)
- Chloromethane (CH₃Cl)
1. Methane (CH₄): Methane is a nonpolar molecule, relying solely on weak LDFs for intermolecular attraction. Its low molecular weight further contributes to weak IMFs.
2. Ethanol (CH₃CH₂OH): Ethanol is a polar molecule exhibiting both LDFs and dipole-dipole forces. More importantly, it possesses a hydroxyl (-OH) group, enabling strong hydrogen bonding.
3. Water (H₂O): Similar to ethanol, water is a polar molecule capable of hydrogen bonding. However, the smaller size of water molecules leads to a higher density of hydrogen bonds compared to ethanol.
4. Chloromethane (CH₃Cl): Chloromethane is a polar molecule, exhibiting dipole-dipole forces in addition to LDFs. However, it lacks the hydrogen bonding capability present in ethanol and water.
Conclusion: Based on the strength of IMFs, the order of increasing boiling points would be: Methane < Chloromethane < Ethanol < Water. Water possesses the highest boiling point due to its extensive hydrogen bonding network. Ethanol also exhibits hydrogen bonding, but the density of hydrogen bonds is lower due to its larger size compared to water. Chloromethane, being polar, has stronger IMFs than methane, but weaker than those in molecules with hydrogen bonding. Methane, only relying on LDFs, has the lowest boiling point.
Advanced Considerations and Exceptions
While the general principles outlined above are useful for predicting relative boiling points, exceptions can arise. Factors such as branching and steric hindrance can influence IMF strength. For instance, a branched alkane will typically have a lower boiling point than its straight-chain isomer due to reduced surface area for LDF interactions. Similarly, the presence of specific functional groups can affect the balance of different IMFs.
Furthermore, the role of critical temperature and pressure adds another layer of complexity to the discussion of boiling points. These properties are inherently linked to the intermolecular forces at play and influence the conditions under which the transition from liquid to gas occurs. While typically not considered in basic comparisons, a deeper understanding of critical parameters is vital for advanced applications and analyses.
Practical Applications and Further Research
Understanding the relationship between molecular structure and boiling point has far-reaching implications in various fields. In chemistry, it allows us to predict the physical properties of molecules and design materials with specific boiling points. In chemical engineering, knowledge of boiling points is critical for designing separation processes such as distillation. In pharmaceuticals, the boiling point of a compound is an important parameter to consider during drug development and formulation. Finally, understanding boiling points is crucial in atmospheric science, allowing for the prediction of weather patterns and the study of atmospheric chemistry.
Future research in this area could involve exploring the effects of external factors like pressure and magnetic fields on boiling points. Additionally, the development of more sophisticated computational models to accurately predict boiling points from molecular structures remains an active area of research, promising to contribute to a deeper understanding of the complex interplay of intermolecular forces. Investigating the boiling points of complex molecules, such as polymers and biological macromolecules, remains an ongoing challenge.
The discussion above provides a comprehensive overview of the factors influencing the boiling point of molecules. By understanding the different types of intermolecular forces and their relative strengths, one can accurately predict and interpret boiling points for a wide variety of chemical compounds. Further exploration of advanced concepts and ongoing research will continuously refine our understanding of this fundamental property.
Latest Posts
Latest Posts
-
Electric Field Of A Cylindrical Shell
Apr 06, 2025
-
Predict The Major Products For Each Of The Following Reactions
Apr 06, 2025
-
A Symbiotic Relationship In Which Both Organisms Benefit
Apr 06, 2025
-
Dynamic Equilibrium Is Maintained By The
Apr 06, 2025
-
Which Layer Carries Out Photosynthesis In A Leaf
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Molecules Has The Highest Boiling Point . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
