Which Of The Following Is An Isoelectronic Series
News Leon
Apr 05, 2025 · 6 min read
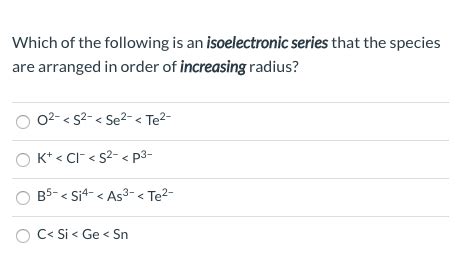
Table of Contents
Which of the Following is an Isoelectronic Series? A Deep Dive into Atomic Structure and Electron Configuration
Understanding isoelectronic series is crucial for grasping fundamental concepts in chemistry, particularly atomic structure and periodic trends. This article will explore what constitutes an isoelectronic series, delve into the underlying principles, and provide numerous examples to solidify your understanding. We'll also explore how this concept relates to other important chemical properties and concepts.
What is an Isoelectronic Series?
An isoelectronic series is a group of atoms, ions, or molecules that have the same number of electrons and the same electronic configuration. Crucially, it's not simply about having the same number of electrons; the arrangement of those electrons in orbitals must also be identical. This identical electronic structure leads to similarities in their chemical and physical properties, although differences in nuclear charge will still affect their properties to some degree.
Think of it like this: imagine building with LEGOs. You have the same number and type of LEGO bricks, but you can arrange them to build different structures. In an isoelectronic series, you're using the same number of "electronic bricks" (electrons), but the "structures" (atoms/ions) are different due to varying nuclear charges.
Identifying Isoelectronic Series: Key Considerations
Identifying an isoelectronic series requires careful attention to the following:
-
Electron Configuration: Determine the electron configuration of each atom or ion in the given set. Remember to account for the charge when dealing with ions; a positive charge means electrons are removed, while a negative charge signifies added electrons. The noble gas configuration provides a shorthand method for representing the electron arrangement.
-
Number of Electrons: Directly compare the total number of electrons in each species. If they differ, they cannot form an isoelectronic series.
-
Electronic Structure: While having the same number of electrons is necessary, it's equally important that the electron shells and subshells are occupied in the same manner. An identical electron configuration is paramount.
Examples of Isoelectronic Series
Let's illustrate this with some examples:
Example 1: A Simple Isoelectronic Series
Consider the following species: O²⁻, F⁻, Ne, Na⁺, Mg²⁺
-
O²⁻: Oxygen gains two electrons to achieve a noble gas configuration, resulting in 10 electrons (1s²2s²2p⁶).
-
F⁻: Fluorine gains one electron to achieve a noble gas configuration, resulting in 10 electrons (1s²2s²2p⁶).
-
Ne: Neon, a noble gas, naturally has 10 electrons (1s²2s²2p⁶).
-
Na⁺: Sodium loses one electron to achieve a noble gas configuration, resulting in 10 electrons (1s²2s²2p⁶).
-
Mg²⁺: Magnesium loses two electrons to achieve a noble gas configuration, resulting in 10 electrons (1s²2s²2p⁶).
All these species have 10 electrons and the same electron configuration (1s²2s²2p⁶). Therefore, they constitute an isoelectronic series. Note that despite their identical electron configurations, their properties will differ due to the varying nuclear charges. For instance, Mg²⁺ will have a smaller ionic radius than O²⁻ due to the greater nuclear charge attracting the electrons more strongly.
Example 2: A More Complex Isoelectronic Series
Let's analyze a more complex set: Cl⁻, Ar, K⁺, Ca²⁺, Sc³⁺
-
Cl⁻: Chlorine gains one electron (18 electrons, 1s²2s²2p⁶3s²3p⁶)
-
Ar: Argon has 18 electrons naturally (1s²2s²2p⁶3s²3p⁶)
-
K⁺: Potassium loses one electron (18 electrons, 1s²2s²2p⁶3s²3p⁶)
-
Ca²⁺: Calcium loses two electrons (18 electrons, 1s²2s²2p⁶3s²3p⁶)
-
Sc³⁺: Scandium loses three electrons (18 electrons, 1s²2s²2p⁶3s²3p⁶)
This set also forms an isoelectronic series because all species have 18 electrons and the same electron configuration. Again, differences in nuclear charge lead to variations in their properties, such as ionic radii and ionization energies.
Example 3: Illustrating a Non-Isoelectronic Series
To highlight the importance of identical electronic configuration, let's consider a non-isoelectronic example: Na⁺, Mg²⁺, Al³⁺, O²⁻
While Na⁺, Mg²⁺, and Al³⁺ all have 10 electrons, O²⁻ possesses 10 electrons as well. However, they do not form an isoelectronic series because their electron configurations differ. Na⁺, Mg²⁺, and Al³⁺ would have a neon-like configuration (1s²2s²2p⁶). O²⁻, while having 10 electrons, would have an electron configuration distinct from the neon core.
Example 4: Isoelectronic Series in Molecules
Isoelectronic series are not limited to atoms and ions; molecules can also be part of such series. For example: CO, N₂, and CN⁻ all possess 14 electrons and a similar electronic structure.
Implications and Applications of Isoelectronic Series
Understanding isoelectronic series has several important implications in various areas of chemistry:
-
Predicting Properties: Isoelectronic species exhibit similar chemical and physical properties, although these are influenced by the nuclear charge. The trend is that for a series of isoelectronic anions, the size will increase with decreasing nuclear charge, whilst for cations, the reverse is true. This allows for the prediction of properties of one species based on the known properties of others within the series.
-
Ionic Radii Trends: By comparing ionic radii within an isoelectronic series, one can gain insights into the effects of nuclear charge on atomic size.
-
Ionization Energies: Similarly, analyzing ionization energies within an isoelectronic series provides a detailed understanding of the relationship between nuclear charge and electron removal energies.
-
Spectroscopic Analysis: Isoelectronic species often show similar spectroscopic patterns due to the resemblance in their electron configurations.
-
Computational Chemistry: Isoelectronic series are frequently used in computational chemistry for benchmarking and validation of theoretical models.
Distinguishing Isoelectronic Series from Other Chemical Concepts
It's crucial to differentiate isoelectronic series from other related concepts:
-
Isotopes: Isotopes are atoms of the same element with the same number of protons but a different number of neutrons. They have different mass numbers but the same electron configuration.
-
Isomers: Isomers are molecules with the same molecular formula but different structural arrangements. They may not be isoelectronic.
-
Isobars: Isobars are atoms of different elements with the same mass number but different atomic numbers.
-
Homologous series: This term is primarily used in organic chemistry for compounds with a similar structure and functional group. They may or may not be isoelectronic.
Conclusion: The Significance of Isoelectronic Series
Isoelectronic series offers a powerful tool for understanding the relationship between electronic structure, nuclear charge, and the properties of atoms and ions. By recognizing the similarities and differences within these series, chemists gain valuable insights into the behaviour of matter at the atomic and molecular levels. The concept extends beyond simple atomic structures, influencing various fields of chemical research and application. Mastering this concept is essential for any student or professional working in chemistry. From predicting properties to understanding spectroscopic data, the principle of isoelectronic series is a fundamental cornerstone of chemical knowledge. Through continued exploration and application, our comprehension of this crucial aspect of atomic structure will only continue to deepen.
Latest Posts
Latest Posts
-
Sequence Of Events In Muscle Contraction
Apr 05, 2025
-
What Is The End Product Of The Calvin Cycle
Apr 05, 2025
-
Which Of The Following Is Not A Solid
Apr 05, 2025
-
The Function Of Dna Ligase In Recombinant Technology Is To
Apr 05, 2025
-
Which Term Refers To A Nations Transportation And Communication Networks
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is An Isoelectronic Series . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
