Which Of The Following Is A Property Of A Solid
News Leon
Apr 02, 2025 · 6 min read
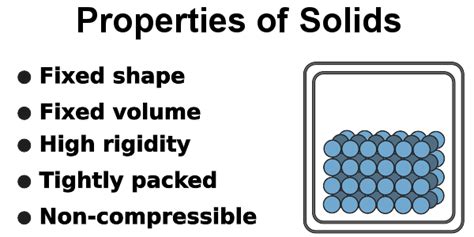
Table of Contents
Which of the Following is a Property of a Solid? Exploring the Defining Characteristics of Solids
The question, "Which of the following is a property of a solid?" might seem simple at first glance. However, a deep understanding of the properties of solids requires exploring the intricate world of matter at the atomic and molecular level. Solids, unlike liquids and gases, possess unique characteristics that define their behavior and interactions. This article delves into the key properties of solids, differentiating them from other states of matter and providing a comprehensive understanding of their defining characteristics. We will explore various properties, clarifying which ones are definitive and which are merely characteristic of many, but not all, solids.
Defining Solids: A Microscopic Perspective
Before we dive into the specific properties, it's crucial to understand the fundamental nature of a solid. At a microscopic level, solids are characterized by strong intermolecular forces holding their constituent particles (atoms, ions, or molecules) in a fixed, rigid structure. These particles are closely packed together, resulting in a definite shape and volume. This fixed arrangement is the primary reason behind many of the observable properties of solids. The strength of these intermolecular forces dictates the overall properties of the solid, influencing its hardness, melting point, and other characteristics.
Key Properties of Solids: A Detailed Exploration
Several properties distinguish solids from liquids and gases. Let's explore some of the most important ones:
1. Definite Shape and Volume: The Hallmark of Solids
This is arguably the most defining property of a solid. Unlike liquids, which take the shape of their container, and gases, which expand to fill the available volume, solids maintain a fixed shape and volume regardless of their container. This is a direct consequence of the strong intermolecular forces holding the constituent particles in a rigid, ordered arrangement. The particles are not free to move around; instead, they vibrate around their fixed positions.
2. Incompressibility: Resistance to Compression
Solids are generally incompressible, meaning their volume doesn't change significantly even under significant pressure. This is because the particles are already tightly packed together, leaving minimal space for further compression. While some compression might occur at extremely high pressures, the change in volume is usually negligible compared to liquids or gases. This property is directly related to the strong intermolecular forces and the rigid structure of solids.
3. High Density: Packing Efficiency
Due to the close packing of particles, solids generally possess high density compared to liquids and gases. The density of a solid depends on the mass of its constituent particles and how efficiently they are packed together. Different crystal structures can result in variations in density, even for the same material. For instance, the density of diamond (a crystalline form of carbon) is significantly higher than that of graphite (another form of carbon).
4. Rigidity and Hardness: Resistance to Deformation
The strong intermolecular forces in solids contribute to their rigidity and hardness. Solids resist deformation under stress, meaning they don't readily change shape when subjected to external forces. However, the degree of rigidity and hardness varies significantly depending on the type of solid and the strength of its intermolecular forces. Some solids, like diamond, are extremely hard and resistant to scratching, while others are relatively soft and easily deformable. This variation highlights the diverse nature of solids and the range of intermolecular forces that can exist.
5. Low Rate of Diffusion: Limited Particle Movement
Unlike liquids and gases where particles move freely, the movement of particles in solids is highly restricted. Diffusion in solids is a slow process because the particles are essentially fixed in their positions and can only vibrate around their equilibrium points. While some atomic-level movement does occur, it is significantly slower than in liquids or gases. This slow diffusion is a key characteristic that distinguishes solids from other states of matter.
6. Crystalline vs. Amorphous Solids: Structural Variations
While the properties mentioned above are common to most solids, it's important to distinguish between crystalline and amorphous solids. Crystalline solids have a highly ordered, repeating three-dimensional arrangement of particles, forming a crystal lattice. This ordered structure contributes to their anisotropy – meaning their properties may vary depending on the direction. Examples include diamonds, salt, and quartz. Amorphous solids, on the other hand, lack this long-range order; their particles are arranged randomly. Glass and rubber are examples of amorphous solids, they are isotropic, meaning their properties are the same in all directions. Even though they share some similar properties like fixed shape and volume, their structural differences lead to variations in other properties such as melting point and optical behavior.
7. Melting Point: The Transition to Liquid State
Solids have a well-defined melting point, the temperature at which they transition to the liquid state. At this temperature, the kinetic energy of the particles overcomes the intermolecular forces holding them in the rigid structure, allowing them to move more freely. The melting point is a crucial property used to identify and characterize different solids. It depends on the strength of intermolecular forces; stronger forces result in higher melting points.
8. Thermal Expansion: Sensitivity to Temperature
Solids, like other states of matter, undergo thermal expansion, meaning their volume increases with increasing temperature. This is because the increased kinetic energy of the particles causes them to vibrate more vigorously, slightly increasing the average distance between them. The extent of thermal expansion varies depending on the material and its structure. This property has important implications in engineering and construction, where thermal expansion and contraction need to be accounted for.
Properties That Aren't Always Definitive of Solids: Important Considerations
While the properties discussed above are generally characteristic of solids, it's crucial to note that some properties are not universally applicable. For instance:
-
Brittleness: While many solids are brittle, meaning they fracture easily under stress, others are ductile (capable of being drawn into wires) or malleable (capable of being hammered into sheets). This depends on the nature of the intermolecular forces and the crystal structure.
-
Elasticity: Some solids exhibit elasticity, meaning they can return to their original shape after deformation. However, this is not a universal property; many solids undergo permanent deformation under stress.
-
Electrical Conductivity: Solids can be either conductors, insulators, or semiconductors of electricity, depending on their electronic structure. This property is not directly related to the solid state itself but rather to the nature of the material's constituent particles.
Conclusion: Understanding the Nuances of Solid Properties
In conclusion, while a definite shape and volume are the most defining properties of a solid, understanding the interplay of intermolecular forces, crystal structure, and other factors is crucial for a comprehensive appreciation of their diverse properties. The rigidity, incompressibility, high density, low diffusion rate, and well-defined melting point are all significant characteristics that help distinguish solids from liquids and gases. However, it's essential to recognize that not all solids share every property to the same extent. The variations in properties highlight the rich complexity of the solid state and underscore the importance of considering the specific characteristics of individual solids when analyzing their behavior and applications. This comprehensive understanding is essential across diverse fields, from material science and engineering to chemistry and physics.
Latest Posts
Latest Posts
-
Which Of The Following Is Not A Transition Metal
Apr 03, 2025
-
When Air Is Heated What Happens
Apr 03, 2025
-
Find X So That L Is Parallel To M
Apr 03, 2025
-
A Price Floor Set Above The Equilibrium Price Will
Apr 03, 2025
-
Genes For The Resistance Of Antibiotics Are Often Located
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is A Property Of A Solid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
