Which Of The Following Ground State Electron Configuration
News Leon
Apr 02, 2025 · 6 min read
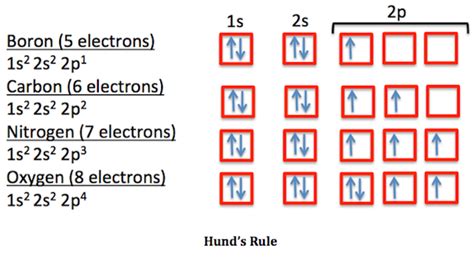
Table of Contents
Decoding Ground State Electron Configurations: A Deep Dive into Atomic Structure
Understanding ground state electron configurations is fundamental to comprehending the behavior of atoms and the periodic trends observed in chemical properties. This article will delve into the principles governing electron configuration, exploring how we determine the ground state, the exceptions to the rules, and the implications for chemical reactivity. We'll also examine how understanding electron configuration helps explain various phenomena in chemistry and physics.
What is an Electron Configuration?
An electron configuration describes the arrangement of electrons within the electron shells and subshells of an atom. Each electron occupies a specific orbital characterized by a set of quantum numbers (principal quantum number, n; azimuthal quantum number, l; magnetic quantum number, ml; and spin quantum number, ms). These quantum numbers dictate the energy level, shape, spatial orientation, and spin of the electron.
The principal quantum number (n) defines the energy level or shell, with n = 1 representing the lowest energy level and increasing values corresponding to higher energy levels. The azimuthal quantum number (l) determines the subshell or orbital shape (l = 0 for s orbitals, l = 1 for p orbitals, l = 2 for d orbitals, l = 3 for f orbitals, and so on). The magnetic quantum number (ml) specifies the orientation of the orbital in space, and the spin quantum number (ms) represents the intrinsic angular momentum of the electron (either +1/2 or -1/2, often represented as ↑ and ↓).
Building Up Principle (Aufbau Principle) and Hund's Rule
Determining the ground state electron configuration involves following specific rules:
-
Aufbau Principle: Electrons fill atomic orbitals in order of increasing energy. This generally follows the order 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p… Note that the order isn't strictly sequential due to orbital energy level crossings. For example, the 4s subshell is lower in energy than the 3d subshell.
-
Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers. This means each orbital can hold a maximum of two electrons, with opposite spins.
-
Hund's Rule: When filling degenerate orbitals (orbitals with the same energy), electrons will singly occupy each orbital within a subshell before pairing up. This maximizes the total spin and results in a more stable configuration.
Writing Electron Configurations: Examples
Let's illustrate electron configuration notation with examples:
-
Hydrogen (H): 1s¹ Hydrogen has one electron, which fills the 1s orbital.
-
Helium (He): 1s² Helium has two electrons, both filling the 1s orbital with opposite spins.
-
Lithium (Li): 1s²2s¹ Lithium has three electrons; two fill the 1s orbital, and the third occupies the 2s orbital.
-
Carbon (C): 1s²2s²2p² Carbon has six electrons. The first four fill the 1s and 2s orbitals, and the remaining two occupy separate 2p orbitals (following Hund's rule).
-
Oxygen (O): 1s²2s²2p⁴ Oxygen has eight electrons; two occupy each of the 2s and 2p orbitals, with the last two p-orbitals having paired electrons.
Exceptions to the Rules: Why Some Elements Don't Follow the Aufbau Principle
While the Aufbau principle provides a good approximation, several elements deviate from the predicted electron configuration. These exceptions usually involve partially filled or nearly filled d or f subshells. The energy difference between these subshells can be small enough that electronic interactions lead to a more stable configuration than predicted by the simple Aufbau principle.
A classic example is Chromium (Cr): [Ar] 3d⁵4s¹ instead of the expected [Ar] 3d⁴4s². The half-filled d subshell (d⁵) and half-filled s subshell (s¹) offer enhanced stability due to exchange energy, which arises from the interactions between electrons with parallel spins. Similar exceptions are observed in copper (Cu) and other transition metals.
Another example is Copper (Cu): [Ar] 3d¹⁰4s¹ instead of [Ar] 3d⁹4s². Here, a completely filled d subshell (d¹⁰) provides additional stability.
These exceptions highlight the limitations of simple rules and underscore the importance of considering electron-electron interactions when predicting ground state configurations. The most accurate configurations are obtained through complex quantum mechanical calculations.
Noble Gas Notation (Shorthand Notation)
To simplify writing long electron configurations, noble gas notation is used. This involves representing the core electrons with the symbol of the preceding noble gas in brackets.
For example:
-
Sodium (Na): [Ne] 3s¹ instead of 1s²2s²2p⁶3s¹
-
Iron (Fe): [Ar] 3d⁶4s² instead of 1s²2s²2p⁶3s²3p⁶3d⁶4s²
Implications of Electron Configuration for Chemical Properties
The electron configuration of an atom significantly influences its chemical properties, particularly its reactivity.
-
Valence Electrons: The electrons in the outermost shell (valence electrons) are primarily responsible for chemical bonding. Atoms tend to react in ways that achieve a stable electron configuration, often resembling that of a noble gas (with a filled outermost shell). This underlies the octet rule, although there are exceptions, particularly for transition metals.
-
Ionization Energy: The energy required to remove an electron from an atom is influenced by its electron configuration. Atoms with stable electron configurations have higher ionization energies.
-
Electron Affinity: The energy change when an electron is added to a neutral atom is also related to the electron configuration. Atoms that can achieve a stable configuration by gaining an electron generally exhibit high electron affinities.
Electron Configuration and the Periodic Table
The periodic table is organized to reflect the electron configurations of elements. Elements in the same group (vertical column) have similar valence electron configurations, leading to similar chemical properties. For instance, alkali metals (Group 1) all have one valence electron (ns¹), while halogens (Group 17) all have seven valence electrons (ns²np⁵).
Advanced Topics: Electron Configurations and Spectroscopy
Electron configurations play a crucial role in understanding atomic spectroscopy. When atoms absorb energy, electrons can jump to higher energy levels (excited states). When these excited electrons return to their ground state, they emit photons of specific energies, corresponding to the energy difference between the levels. The analysis of these spectral lines provides direct evidence for the existence of specific energy levels and the validity of electron configurations.
Determining Electron Configurations: Tools and Techniques
While the Aufbau principle and Hund's rule provide a framework for predicting electron configurations, more sophisticated methods are necessary for precise determination. Computational chemistry utilizes quantum mechanics to calculate the exact electron configurations of atoms and molecules. These calculations are crucial for understanding the electronic structure of complex systems.
Conclusion: The Importance of Understanding Electron Configurations
Understanding electron configurations is paramount for grasping the fundamental principles of chemistry and physics. It provides a framework for interpreting atomic properties, predicting chemical reactivity, explaining spectroscopic data, and connecting the macroscopic properties of substances to the microscopic world of atoms and their electrons. From simple introductory concepts to advanced computational techniques, the study of electron configurations remains a central theme in the scientific understanding of matter. The exceptions and nuances we've explored highlight the rich complexity of atomic structure and the ongoing pursuit of a deeper understanding of the fundamental laws governing the universe.
Latest Posts
Latest Posts
-
What Is The Molar Mass Of Phosphoric Acid
Apr 03, 2025
-
What Type Of Joint Is In The Skull
Apr 03, 2025
-
Which Of The Following Is An Implicit Cost Of Production
Apr 03, 2025
-
Select The Sentence That Is Punctuated Correctly
Apr 03, 2025
-
Which Of The Following Is The Correct Accounting Equation
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Ground State Electron Configuration . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
