When Two Things Are The Same Temperature
News Leon
Apr 03, 2025 · 7 min read
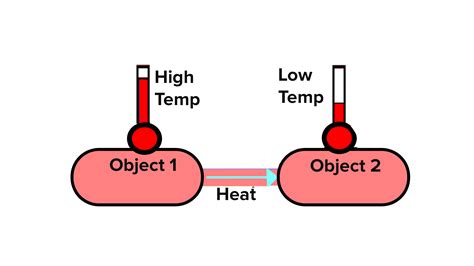
Table of Contents
When Two Things Are the Same Temperature: Exploring Thermal Equilibrium
Understanding temperature is fundamental to comprehending the world around us. From the simmering heat of a stovetop to the icy chill of winter, temperature dictates how we interact with our environment and how various processes unfold. But what truly defines temperature, and what happens when two objects reach the same temperature? This article delves deep into the concept of thermal equilibrium, exploring its implications across various scientific disciplines.
What is Temperature?
Before we explore the implications of two things having the same temperature, we need a solid understanding of what temperature actually is. Temperature isn't a measure of the total energy contained within an object (that's heat), but rather a measure of the average kinetic energy of its constituent particles—atoms and molecules. These particles are constantly in motion, vibrating, rotating, and translating. The faster they move, the higher the average kinetic energy, and consequently, the higher the temperature.
Microscopic View of Temperature
Imagine a container filled with gas molecules. These molecules are zipping around at various speeds, colliding with each other and the container walls. Temperature reflects the average speed of these molecules. A higher temperature indicates a higher average speed, while a lower temperature signifies a slower average speed. It's crucial to remember that even at absolute zero (-273.15°C or 0 Kelvin), particles still possess some vibrational energy, though their average kinetic energy is minimal.
Macroscopic Measurement of Temperature
While the microscopic view helps us understand the underlying physics, we measure temperature macroscopically using various instruments like thermometers. Thermometers utilize the principle of thermal expansion. Most substances expand when heated and contract when cooled. The change in volume is then calibrated to a temperature scale, such as Celsius, Fahrenheit, or Kelvin. Different types of thermometers exploit different properties of matter, such as the expansion of mercury in a glass tube or the change in electrical resistance of a thermistor.
Thermal Equilibrium: Reaching a Consensus
When two objects are placed in contact, a process of energy transfer occurs until they reach the same temperature. This state of thermal balance is called thermal equilibrium. The energy transfer is driven by the difference in temperature between the two objects. Heat, a form of energy, flows spontaneously from the hotter object to the colder object until their temperatures equalize.
Mechanisms of Heat Transfer
Several mechanisms facilitate this heat transfer:
-
Conduction: This is the transfer of heat through direct contact. For example, if you touch a hot stove, heat is conducted from the stove to your hand. The rate of conduction depends on the thermal conductivity of the materials involved—metals are typically good conductors, while insulators like wood are poor conductors.
-
Convection: This involves the transfer of heat through the movement of fluids (liquids or gases). Hotter, less dense fluid rises, while cooler, denser fluid sinks, creating convection currents. This is how a radiator heats a room—hot air rises, circulates, and distributes heat.
-
Radiation: This is the transfer of heat through electromagnetic waves, requiring no medium for transmission. The sun heats the Earth through radiation. All objects emit radiation, and the amount emitted depends on their temperature. This is why even in a vacuum, heat transfer can still occur through radiation.
The Zeroth Law of Thermodynamics
The concept of thermal equilibrium is formally enshrined in the Zeroth Law of Thermodynamics. This law states that if two systems are each in thermal equilibrium with a third system, then they are in thermal equilibrium with each other. This seemingly obvious statement is fundamental because it allows us to use thermometers to measure temperature reliably. The thermometer acts as the third system, and its equilibrium with the object being measured allows for a consistent temperature reading.
Implications of Thermal Equilibrium
The concept of thermal equilibrium has profound implications across various fields:
Engineering and Technology
Thermal management is crucial in many engineering applications. For example, designing efficient engines requires careful consideration of heat transfer and thermal equilibrium to maximize efficiency and minimize waste heat. Similarly, electronics rely on efficient cooling systems to prevent overheating and component failure. Understanding thermal equilibrium allows engineers to design systems that operate within optimal temperature ranges.
Meteorology and Climatology
Thermal equilibrium plays a pivotal role in weather patterns and climate change. The Earth's temperature is a result of a dynamic equilibrium between incoming solar radiation and outgoing terrestrial radiation. Factors like greenhouse gases can disrupt this equilibrium, leading to global warming. Climate models rely heavily on accurate representations of heat transfer processes and the concept of thermal equilibrium to predict future climate scenarios.
Biology and Medicine
Biological systems are remarkably sensitive to temperature. Maintaining a stable internal temperature (homeostasis) is crucial for the proper functioning of organisms. Understanding thermal equilibrium helps us comprehend how organisms regulate their body temperature and adapt to different environmental conditions. In medicine, thermal imaging techniques exploit differences in temperature to diagnose various conditions and monitor treatment responses.
Chemistry and Physics
In chemistry, thermal equilibrium is fundamental to understanding chemical reactions and phase transitions. Reactions often occur more rapidly at higher temperatures, and the equilibrium constant of a reversible reaction is temperature-dependent. In physics, the study of thermal equilibrium is crucial for understanding thermodynamics, statistical mechanics, and the behavior of materials at different temperatures.
Achieving and Maintaining Thermal Equilibrium: Practical Examples
Let's consider some real-world examples of how thermal equilibrium is achieved and maintained:
-
Heating a cup of tea: When you pour hot water into a mug, the hot water is initially at a higher temperature than the mug and the surrounding air. Heat transfers from the water to the mug and the air until thermal equilibrium is reached, and all components are at the same temperature.
-
Cooling a bottle of soda: A warm bottle of soda placed in a refrigerator will gradually cool down as heat transfers from the soda to the cooler air inside the refrigerator, eventually reaching thermal equilibrium with the refrigerator's temperature.
-
Cooking food: Cooking involves carefully controlling the heat transfer to food to achieve a desired final temperature. The food initially starts at room temperature and absorbs heat from a stovetop, oven, or microwave until it reaches the desired cooking temperature.
-
Human Body Temperature Regulation: Our bodies maintain a relatively constant internal temperature through a complex interplay of physiological mechanisms. When our external environment is hotter or colder, our bodies adjust through sweating, shivering, and other processes to maintain thermal equilibrium and prevent overheating or hypothermia.
Beyond Simple Systems: Complexity and Challenges
While the concept of thermal equilibrium is relatively straightforward in simple systems, it becomes significantly more complex in larger and more dynamic systems. For instance, accurately modeling global climate change requires considering countless variables and interactions, making it a complex challenge in achieving and maintaining a stable thermal equilibrium for the Earth's climate system.
The study of thermal equilibrium is an ongoing area of research, with scientists continually striving to better understand the intricate processes governing heat transfer and the conditions that lead to thermal balance in various contexts. From the microscopic interactions of particles to macroscopic climate patterns, thermal equilibrium is an integral concept that impacts various facets of our world.
Conclusion: The Universal Language of Temperature
Ultimately, understanding when two things are at the same temperature, meaning they are in thermal equilibrium, allows us to unravel the complexities of energy transfer and the behavior of matter at various scales. This principle underpins numerous scientific disciplines and technological advancements, making it a cornerstone of our understanding of the physical world. As we continue to explore the universe and its intricacies, the principle of thermal equilibrium remains a fundamental concept that governs the behaviour of everything from subatomic particles to planetary systems. Further research and advancements will undoubtedly deepen our understanding of this crucial concept and its far-reaching consequences.
Latest Posts
Latest Posts
-
Which Atomic Particle Determines The Chemical Behavior Of An Atom
Apr 03, 2025
-
Which Of The Following Occurs First Causing Expiration
Apr 03, 2025
-
A Homogeneous Mixture Is Also Called A
Apr 03, 2025
-
Least Common Multiple Of 3 6 9
Apr 03, 2025
-
Give The Iupac Names For The Following Compounds
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about When Two Things Are The Same Temperature . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
