What Happens When Gas Is Heated
News Leon
Apr 02, 2025 · 6 min read
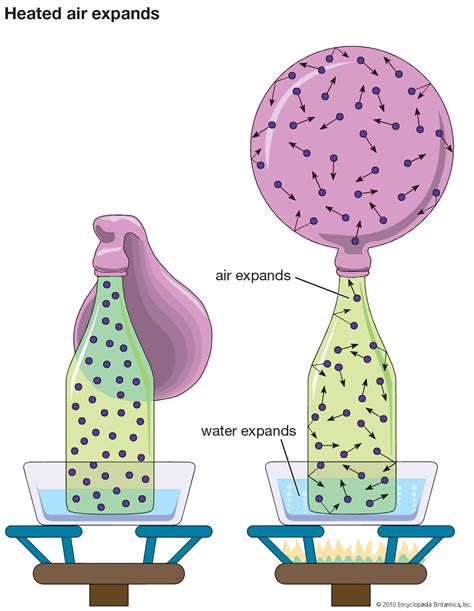
Table of Contents
What Happens When Gas is Heated: A Deep Dive into the Thermodynamics of Gases
The seemingly simple act of heating a gas belies a complex interplay of physical processes governed by the laws of thermodynamics. Understanding what happens when gas is heated is crucial in numerous fields, from engineering and meteorology to chemistry and astrophysics. This comprehensive article will explore the various effects of heating gases, examining the changes in their pressure, volume, temperature, and molecular behavior. We’ll delve into the underlying principles and explore practical applications.
The Fundamental Principles: Gas Laws and Kinetic Theory
Before exploring the effects of heating, it's essential to understand the fundamental principles governing gas behavior. These principles are primarily embodied in the gas laws, which describe the relationships between pressure (P), volume (V), temperature (T), and the amount of gas (n).
The Ideal Gas Law: A Simplified Model
The most fundamental gas law is the ideal gas law, which states:
PV = nRT
Where:
- P is the pressure of the gas
- V is the volume of the gas
- n is the number of moles of gas
- R is the ideal gas constant
- T is the absolute temperature of the gas (in Kelvin)
This law assumes that gas molecules are point masses with negligible volume and that there are no intermolecular forces between them. While this is a simplification, the ideal gas law provides a good approximation for many gases under normal conditions.
Kinetic Theory of Gases: The Microscopic Perspective
The kinetic theory of gases provides a microscopic explanation for the macroscopic behavior described by the gas laws. It states that gases consist of a large number of tiny particles (atoms or molecules) that are in constant, random motion. The temperature of a gas is directly proportional to the average kinetic energy of these particles.
Heating a gas increases the average kinetic energy of its molecules. This increased kinetic energy manifests in several ways, leading to observable changes in the gas's macroscopic properties.
The Effects of Heating a Gas: A Multifaceted Transformation
Heating a gas leads to a cascade of effects, each intricately linked to the others. Let's explore these effects in detail:
1. Increase in Temperature: The Direct Effect
The most immediate and obvious effect of heating a gas is an increase in its temperature. This is because heat energy is transferred to the gas molecules, increasing their average kinetic energy. The temperature increase is directly proportional to the amount of heat added, assuming no work is done by the gas.
2. Expansion: Volume Changes at Constant Pressure
If the gas is allowed to expand freely (i.e., at constant pressure), heating it will cause an increase in its volume. This is because the increased kinetic energy of the molecules leads to more frequent and forceful collisions with the container walls, pushing the walls outwards and increasing the volume. This is essentially Charles's Law, which states that at constant pressure, the volume of a gas is directly proportional to its absolute temperature.
3. Pressure Increase: Volume Changes at Constant Volume
If the gas is confined to a fixed volume (i.e., at constant volume), heating it will cause an increase in its pressure. The increased kinetic energy of the molecules leads to more frequent and forceful collisions with the container walls, resulting in a higher pressure. This is Gay-Lussac's Law, which states that at constant volume, the pressure of a gas is directly proportional to its absolute temperature.
4. Changes in Density: A Consequence of Volume and Temperature
The density of a gas is defined as its mass per unit volume. Heating a gas generally leads to a decrease in its density, primarily due to the volume expansion at constant pressure. However, the density change depends on the specific conditions, as a pressure increase at constant volume can lead to a relatively small density change.
5. Changes in Molecular Behavior: Beyond the Ideal Gas Model
The ideal gas law provides a good approximation for many gases under normal conditions. However, at high pressures or low temperatures, the assumptions of the ideal gas law break down. Intermolecular forces and the finite volume of the molecules become significant, leading to deviations from ideal behavior. Heating a real gas under such conditions can lead to more complex changes in its properties. For example, the compressibility factor, a measure of deviation from ideal behavior, can change significantly with temperature.
Applications: From Everyday Life to Advanced Technology
The principles of heating gases find widespread applications across diverse fields:
1. Internal Combustion Engines: The Power of Expansion
Internal combustion engines rely on the expansion of heated gases to generate mechanical work. The combustion of fuel within the engine cylinder rapidly increases the temperature and pressure of the gases, causing them to expand and push the piston, thereby producing power.
2. Hot Air Balloons: Buoyancy Through Thermal Expansion
Hot air balloons utilize the principle of thermal expansion. Heating the air inside the balloon decreases its density, making it lighter than the surrounding cooler air and generating buoyant force, allowing the balloon to rise.
3. Meteorology and Climate Science: Atmospheric Dynamics
The heating of the Earth's atmosphere by solar radiation drives atmospheric circulation patterns, weather systems, and climate dynamics. Understanding the effects of heating gases is crucial for accurate weather forecasting and climate modeling.
4. Industrial Processes: Chemical Reactions and Manufacturing
Many industrial processes involve heating gases to drive chemical reactions or to modify the properties of materials. Examples include the production of ammonia via the Haber-Bosch process and various metallurgical processes involving heating gases to refine metals.
5. Aerospace Engineering: Rocket Propulsion and Aerodynamics
Rocket propulsion relies on the rapid expansion of heated gases expelled from a nozzle to generate thrust. Understanding the behavior of heated gases is essential for designing efficient and safe rocket engines. Aerodynamics also involves understanding the behavior of gases around aircraft and spacecraft, which is significantly affected by temperature changes.
Beyond the Basics: Advanced Concepts
The effects of heating gases can be further explored using more advanced concepts:
1. Specific Heat Capacity: A Measure of Heat Absorption
The specific heat capacity of a gas represents the amount of heat required to raise the temperature of one unit mass of the gas by one degree. Different gases have different specific heat capacities, reflecting variations in their molecular structures and interactions.
2. Adiabatic Processes: No Heat Exchange
An adiabatic process is one where no heat exchange occurs between the gas and its surroundings. During an adiabatic expansion, the gas cools as it does work against its surroundings, while during an adiabatic compression, the gas heats up.
3. Isothermal Processes: Constant Temperature
An isothermal process is one where the temperature of the gas remains constant. This can be achieved by allowing heat exchange with the surroundings during expansion or compression.
4. Polytropic Processes: Generalized Relationships
Polytropic processes represent a generalization of adiabatic and isothermal processes, describing relationships between pressure and volume that are not necessarily constant temperature or no heat exchange. These processes involve an exponent that characterizes the nature of the process.
Conclusion: A Comprehensive Understanding
Heating a gas leads to a complex interplay of changes in its temperature, pressure, volume, and molecular behavior. Understanding these effects is fundamental to numerous scientific and engineering disciplines. From the everyday workings of internal combustion engines to the complexities of atmospheric dynamics and rocket propulsion, the principles governing heated gases form the bedrock of many critical technologies and scientific phenomena. This deep dive into the subject highlights the intricate relationships between macroscopic observations and the microscopic world of gas molecules, providing a comprehensive understanding of this essential area of physics and chemistry.
Latest Posts
Latest Posts
-
Fill Up The Blanks With The Words Given
Apr 03, 2025
-
Which Of The Following Is True About Tsunamis
Apr 03, 2025
-
Which Of The Following Is Not A Transition Metal
Apr 03, 2025
-
When Air Is Heated What Happens
Apr 03, 2025
-
Find X So That L Is Parallel To M
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Happens When Gas Is Heated . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
