What Chemical Element Has The Highest Electron Affinity
News Leon
Apr 05, 2025 · 5 min read
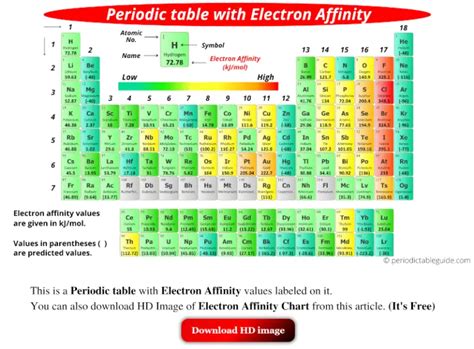
Table of Contents
What Chemical Element Has the Highest Electron Affinity?
Electron affinity, a crucial concept in chemistry and physics, describes the energy change that occurs when an atom gains an electron. Understanding electron affinity is key to predicting chemical behavior and bonding properties. While many elements exhibit a tendency to attract electrons, the question of which element boasts the highest electron affinity is a fascinating and complex one, fraught with subtle nuances and some debate. This article delves deep into the subject, exploring the factors that influence electron affinity, examining the leading contenders, and discussing the challenges in definitively determining the element with the absolute highest electron affinity.
Understanding Electron Affinity: A Deep Dive
Electron affinity (EA) is defined as the energy released when a neutral gaseous atom gains an electron to form a negative ion (anion). A higher (more positive) value of electron affinity indicates a greater tendency for the atom to accept an electron, implying a stronger attraction between the newly acquired electron and the atom's nucleus. It's important to note that the value is often expressed as a negative number in some conventions, representing the energy released, while other conventions use positive numbers. For the purpose of this article, we will focus on the magnitude of the value, with higher values indicating stronger electron affinity.
Several factors influence an element's electron affinity:
1. Effective Nuclear Charge: The Pull of the Nucleus
The effective nuclear charge (Z<sub>eff</sub>) is the net positive charge experienced by an electron in an atom. A higher Z<sub>eff</sub> means a stronger pull from the nucleus, increasing the atom's ability to attract and bind an additional electron. Elements with high Z<sub>eff</sub> generally exhibit high electron affinities.
2. Atomic Size: Distance Matters
The distance between the nucleus and the outermost electron shell significantly impacts electron affinity. Smaller atoms have a shorter distance, resulting in a stronger electrostatic attraction between the nucleus and the incoming electron. Therefore, smaller atoms tend to exhibit higher electron affinities.
3. Electron Shell Configuration: Stability is Key
The stability of the electron configuration plays a critical role. Atoms strive for stable electron configurations, often resembling noble gas configurations (full outer electron shells). Adding an electron to an atom that results in a more stable configuration leads to a higher electron affinity. For example, halogens (Group 17) readily accept an electron to achieve a stable noble gas configuration.
4. Electron-Electron Repulsion: The Crowding Effect
The presence of other electrons in the same electron shell can significantly affect electron affinity. Electron-electron repulsion counteracts the attractive force of the nucleus. Adding an electron to a shell that is already populated with many electrons can lead to increased repulsion and a lower electron affinity.
The Contenders for Highest Electron Affinity: A Detailed Look
While chlorine (Cl) is often cited as having the highest electron affinity, this isn't entirely accurate and requires a more nuanced understanding. The electron affinity values are experimentally determined and can vary slightly depending on the measurement techniques and conditions. Furthermore, the concept of electron affinity becomes more complicated for subsequent electron additions (second electron affinity, third electron affinity, etc.). The first electron affinity is generally the focus of most discussions.
Here's a closer look at some key elements and why their electron affinities are high:
Chlorine (Cl): The Usual Suspect
Chlorine is frequently mentioned as the element with the highest electron affinity. Its high electron affinity stems from its relatively small atomic size and high effective nuclear charge. Adding an electron to chlorine completes its outer electron shell, achieving a stable noble gas configuration, further enhancing its electron affinity.
Fluorine (F): The Unexpected Competitor
Fluorine, though having a smaller atomic size than chlorine, has a slightly lower electron affinity. This seemingly paradoxical result can be attributed to increased electron-electron repulsion in the relatively small 2p subshell of fluorine. The added electron experiences greater repulsion from the already existing electrons in this compact shell, slightly reducing the overall attractive force.
Oxygen (O): The Complicated Case
Oxygen shows a relatively high electron affinity, yet it's lower than that of chlorine and fluorine. Similar to fluorine, oxygen's relatively compact size and electron-electron repulsion in its 2p subshell contribute to a reduced electron affinity compared to chlorine.
The Challenges in Defining the Absolute Highest Electron Affinity
Pinpointing the element with the absolute highest electron affinity is challenging due to several factors:
Experimental Limitations: Measuring the Unmeasurable
Precisely measuring electron affinities can be technically demanding. The values obtained can vary based on experimental techniques and conditions. Small uncertainties in measurements can significantly affect the ranking of elements with closely matched electron affinities.
Theoretical Complications: The Limits of Calculation
Theoretical calculations of electron affinities are subject to inherent approximations and limitations. Accurate predictions require sophisticated computational methods and can still have inherent uncertainties.
Beyond the First Electron Affinity: The Multi-Electron Picture
The discussion of electron affinity often focuses on the first electron affinity (addition of one electron). However, adding subsequent electrons becomes progressively less favorable due to increased electron-electron repulsion. The second electron affinity (adding a second electron) is invariably positive (meaning energy is required, not released), and the third and subsequent electron affinities become increasingly positive.
Conclusion: A Complex Question with No Simple Answer
While chlorine is often cited as having the highest electron affinity, the reality is more nuanced. Fluorine’s smaller size makes it a strong contender, but electron-electron repulsion complicates the picture. The precise ranking of elements based on their electron affinity remains a subject of ongoing research and refinement. The experimental challenges and the theoretical intricacies inherent in determining these values prevent a definitive, universally accepted answer to the question of which element possesses the absolute highest electron affinity. Ultimately, the factors influencing electron affinity are intricate and interconnected, highlighting the complexity of atomic interactions and the ongoing pursuit of precise scientific understanding. Further research and refinements in experimental techniques and theoretical models will continue to enhance our understanding of this fundamental property of chemical elements.
Latest Posts
Latest Posts
-
Correctly Match The Following Joint Symphysis
Apr 06, 2025
-
The Solid Part Of The Earth
Apr 06, 2025
-
How Many Asymmetric Carbons Are Present In The Compound Below
Apr 06, 2025
-
K2 Cr2 O7 H2 S O4
Apr 06, 2025
-
Why The Electric Field Inside A Conductor Is Zero
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Chemical Element Has The Highest Electron Affinity . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
