The Iupac Name Of This Compound Is Methyl Butanoate
News Leon
Apr 02, 2025 · 6 min read
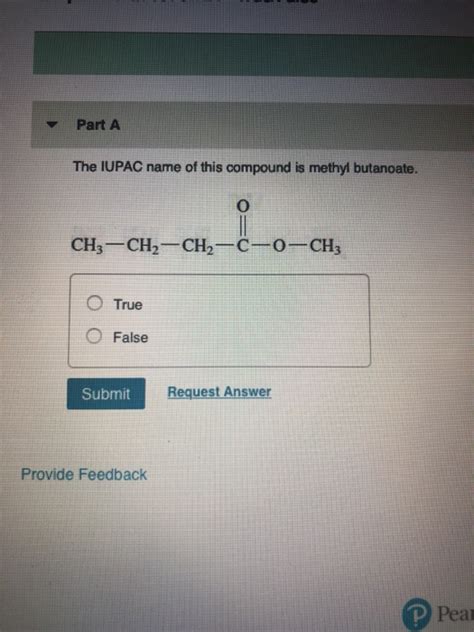
Table of Contents
The IUPAC Name of This Compound is Methyl Butanoate: A Deep Dive into Ester Chemistry
The compound methyl butanoate, also known by its common name methyl butyrate, holds a significant place in the world of organic chemistry and beyond. Its simple structure belies its multifaceted nature, impacting various industries from food flavoring to fragrance manufacturing. This article will delve deeply into the chemical properties, nomenclature, synthesis, applications, and safety considerations of this fascinating ester.
Understanding the IUPAC Nomenclature
The International Union of Pure and Applied Chemistry (IUPAC) provides a standardized system for naming chemical compounds, ensuring clarity and consistency across the scientific community. Methyl butanoate's IUPAC name directly reflects its molecular structure. Let's break it down:
-
Butanoate: This part signifies the carboxylic acid portion of the ester. "Butanoate" indicates a four-carbon chain (but-) with a carboxyl group (-oate) attached. The parent chain is considered butanoic acid (butyric acid).
-
Methyl: This prefix designates the alkyl group attached to the oxygen atom of the carboxyl group. "Methyl" signifies a one-carbon alkyl group (-CH₃).
Therefore, methyl butanoate clearly communicates that the compound is an ester derived from butanoic acid and methanol. The systematic naming system allows chemists worldwide to instantly understand the molecule's structure based solely on its IUPAC name.
Distinguishing IUPAC from Common Names
While IUPAC names offer precision and universality, common names often persist due to historical usage or simplicity. Methyl butanoate's common name, methyl butyrate, is a prime example. "Butyrate" is a shorter, more familiar version of "butanoate," making the common name more convenient in informal settings. However, for formal scientific communication and precise chemical identification, the IUPAC name is always preferred.
Chemical Properties of Methyl Butanoate
Methyl butanoate is a colorless liquid at room temperature, possessing a characteristic fruity, apple-like odor. This characteristic aroma contributes significantly to its widespread use in the flavor and fragrance industries. Its key chemical properties include:
- Molecular Formula: C₅H₁₀O₂
- Molar Mass: 102.13 g/mol
- Density: Approximately 0.90 g/mL
- Boiling Point: Approximately 102 °C
- Solubility: Slightly soluble in water but readily soluble in many organic solvents.
- Flammability: Highly flammable.
Ester Functional Group and its Reactivity
Methyl butanoate's core characteristic stems from its ester functional group (-COO-). This group is responsible for its characteristic reactivity, including:
-
Hydrolysis: Esters can undergo hydrolysis, a reaction with water (often catalyzed by acid or base), to produce the parent carboxylic acid (butanoic acid) and alcohol (methanol).
-
Saponification: Base-catalyzed hydrolysis, specifically with a strong base like sodium hydroxide (NaOH), is called saponification. This process is crucial in soap making and yields the sodium salt of the carboxylic acid and the alcohol.
-
Transesterification: Esters can react with other alcohols in the presence of a catalyst to exchange the alkyl group attached to the oxygen atom. This reaction is vital in biodiesel production.
Synthesis of Methyl Butanoate
Several methods can be employed to synthesize methyl butanoate. The most common and straightforward approach is Fischer esterification. This acid-catalyzed reaction involves the direct esterification of butanoic acid with methanol:
CH₃CH₂CH₂COOH (Butanoic acid) + CH₃OH (Methanol) ⇌ CH₃CH₂CH₂COOCH₃ (Methyl butanoate) + H₂O (Water)
This reaction is typically carried out in the presence of a strong acid catalyst, such as sulfuric acid (H₂SO₄) or p-toluenesulfonic acid (TsOH), to accelerate the equilibrium towards ester formation. Removal of water during the reaction can further drive the equilibrium to the right, maximizing the yield of methyl butanoate.
Alternative Synthetic Routes
While Fischer esterification is the most prevalent method, alternative routes exist. These include:
-
Esterification of butanoyl chloride (butyryl chloride): This method involves reacting butanoyl chloride with methanol. This reaction is more efficient than Fischer esterification because it doesn't involve an equilibrium.
-
Transesterification: This process involves reacting another ester with methanol in the presence of a catalyst. This method can be useful if you have a readily available starting ester.
The choice of synthetic route depends on factors such as the availability of starting materials, desired purity, and scale of production.
Applications of Methyl Butanoate
The versatility of methyl butanoate makes it a valuable compound in numerous industries. Its prominent applications include:
Flavor and Fragrance Industry
Methyl butanoate's appealing fruity aroma is its most significant attribute. It's extensively used as a flavoring agent in various food products, including:
- Artificial fruit flavors: It contributes significantly to the characteristic aroma of apples, pears, and pineapples in processed foods, candies, and beverages.
- Dairy products: It can enhance the flavor profiles of yogurt, ice cream, and other dairy-based products.
- Baked goods: It imparts a fruity note to cakes, pastries, and other baked items.
In the fragrance industry, methyl butanoate adds a pleasant, fruity note to perfumes, colognes, and other personal care products.
Other Applications
Beyond its use in flavor and fragrance, methyl butanoate also finds applications in:
- Solvent: Due to its solubility properties, it can act as a solvent in certain chemical processes.
- Intermediate in chemical synthesis: It can serve as a starting material for the synthesis of other organic compounds.
- Pheromone research: Some insects utilize esters similar to methyl butanoate as pheromones, so it plays a role in pheromone research.
Safety Considerations
While methyl butanoate is generally considered relatively safe, certain precautions are necessary when handling it:
- Flammability: It's highly flammable and should be kept away from open flames and ignition sources.
- Inhalation: Inhalation of its vapors can cause irritation of the respiratory tract. Adequate ventilation is crucial when handling it.
- Skin and Eye Contact: Contact with skin or eyes can cause irritation. Appropriate protective equipment, such as gloves and safety goggles, should be worn.
- Ingestion: Ingestion can cause gastrointestinal irritation. Proper handling and storage are essential to prevent accidental ingestion.
Methyl Butanoate and the Environment
The environmental impact of methyl butanoate is relatively low compared to some other chemicals. However, its disposal should follow appropriate guidelines to prevent pollution. Proper waste management is crucial to minimize its potential impact on the environment. Biodegradability studies have indicated that it is readily biodegradable under appropriate conditions.
Conclusion
Methyl butanoate, with its straightforward IUPAC name reflecting its chemical structure, is much more than just a simple ester. Its pleasant aroma, coupled with its versatile reactivity and various applications, highlight its importance across numerous industries. Understanding its chemical properties, synthesis methods, applications, and safety considerations is crucial for anyone working with this valuable compound. The continued research and development surrounding methyl butanoate and related esters promise further innovations in flavoring, fragrance, and other chemical applications in the future. The precise and standardized naming provided by IUPAC nomenclature remains vital for clear communication and collaboration within the scientific community, ensuring the safe and responsible use of this important chemical.
Latest Posts
Latest Posts
-
Find The Values Of X And Y In Parallelogram Pqrs
Apr 03, 2025
-
Select The Correct Statement About Plant Life Cycles
Apr 03, 2025
-
What Is The Molar Mass Of Phosphoric Acid
Apr 03, 2025
-
What Type Of Joint Is In The Skull
Apr 03, 2025
-
Which Of The Following Is An Implicit Cost Of Production
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Iupac Name Of This Compound Is Methyl Butanoate . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
