Ice Has Lower Density Than Water Because Ice
News Leon
Apr 03, 2025 · 5 min read
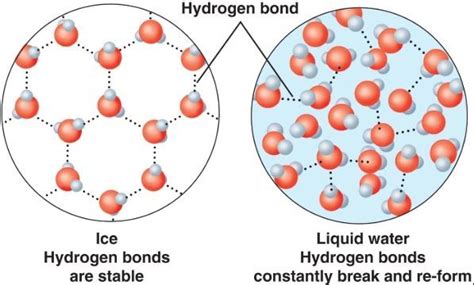
Table of Contents
Ice Has Lower Density Than Water Because Ice… Forms a Crystal Lattice Structure
Water is unique. Unlike most substances, it's denser in its liquid form than in its solid form (ice). This seemingly simple fact has profound implications for life on Earth, shaping our planet's climate, ecosystems, and even the very possibility of aquatic life. But why does ice float? The answer lies in the remarkable structure of water molecules and the way they arrange themselves when freezing.
The Hydrogen Bond: The Key to Water's Anomalous Behavior
The unusual properties of water, including its lower density as ice, stem primarily from the hydrogen bond. A hydrogen bond is a relatively weak attraction between a hydrogen atom in one molecule and an electronegative atom (like oxygen) in another molecule. In water (H₂O), the oxygen atom is more electronegative than the hydrogen atoms, creating a slightly negative charge on the oxygen and slightly positive charges on the hydrogens. This polarity allows water molecules to form hydrogen bonds with up to four neighboring molecules.
Understanding Polarity and Electronegativity
Electronegativity refers to an atom's ability to attract electrons in a chemical bond. Oxygen is highly electronegative, meaning it pulls the shared electrons in the O-H bonds closer to itself. This creates a dipole moment, making the water molecule slightly polar – one end (oxygen) is partially negative (δ-), and the other end (hydrogens) is partially positive (δ+).
This polarity is crucial because it allows water molecules to interact strongly with each other through hydrogen bonding. These bonds are not as strong as covalent bonds (the bonds within the water molecule itself), but they are significant enough to influence the physical properties of water.
The Crystal Lattice Structure of Ice
When water freezes, the hydrogen bonds between molecules become more ordered and stable. Instead of the constantly shifting, chaotic arrangement in liquid water, the molecules organize themselves into a crystalline lattice structure. This structure is characterized by a relatively open, hexagonal arrangement of water molecules.
Visualizing the Hexagonal Lattice
Imagine building a honeycomb structure. The water molecules, in their effort to maximize hydrogen bonding, position themselves at the corners of this structure. The hexagonal gaps create significant empty space within the ice crystal. This open structure is the key to understanding why ice is less dense than liquid water.
Comparing Liquid and Solid Structures
In liquid water, the hydrogen bonds are constantly breaking and reforming, resulting in a more disordered and compact arrangement of molecules. The molecules are closer together, leading to a higher density. In ice, however, the fixed hexagonal structure dictates a more spacious arrangement. The molecules are held farther apart by the rigid hydrogen bonds, resulting in a lower density.
The Implications of Ice's Lower Density
The fact that ice floats has far-reaching consequences for life on Earth and our planet's environment.
Insulating Effect on Aquatic Life
Perhaps the most crucial implication is the insulating effect ice provides for aquatic life. Because ice floats, it forms a layer on the surface of lakes and oceans during winter. This layer acts as an insulating blanket, preventing the water below from freezing solid. This allows aquatic organisms to survive even in extremely cold temperatures. Without this insulating layer, bodies of water would freeze from the bottom up, potentially eliminating most aquatic life.
Moderating Earth's Climate
Ice's lower density also plays a critical role in moderating Earth's climate. Large ice sheets and glaciers reflect a significant portion of incoming solar radiation back into space, contributing to a cooling effect. This albedo effect helps regulate global temperatures. Changes in the extent of ice cover can significantly impact Earth's climate system.
Ice's Role in Shaping Landscapes
The expansion of water upon freezing also has a powerful effect on shaping landscapes. Water seeping into cracks in rocks can freeze and expand, exerting considerable pressure that can break the rocks apart. This process of frost wedging is a significant contributor to weathering and erosion in many environments.
Beyond the Basics: Further Exploration of Water's Anomalous Properties
The lower density of ice compared to liquid water is just one example of water's anomalous behavior. Several other unusual properties result from the unique hydrogen bonding network:
-
High Specific Heat Capacity: Water can absorb a large amount of heat energy with a relatively small temperature change. This makes water an excellent temperature regulator, helping to moderate climate fluctuations.
-
High Heat of Vaporization: Water requires a significant amount of energy to transition from a liquid to a gas (evaporation). This property is essential for sweating and transpiration in plants, both critical cooling mechanisms.
-
High Surface Tension: The strong hydrogen bonds create high surface tension, allowing small insects to walk on water.
-
Universal Solvent: Water's polarity makes it an excellent solvent, able to dissolve many substances. This property is essential for life, as it facilitates the transport of nutrients and waste products in biological systems.
Conclusion: The Significance of a Simple Phenomenon
The seemingly simple fact that ice floats has profound implications for the Earth's environment and the existence of life as we know it. The unique hydrogen bonding capabilities of water molecules, leading to the formation of an open crystalline lattice structure in ice, explain this crucial property. This understanding underscores the extraordinary nature of water and its indispensable role in shaping our planet and sustaining life. Further research continues to explore the intricacies of water's behavior, revealing ever more of its surprising and vital characteristics. The seemingly simple question, "Why does ice float?" opens a door to a deeper understanding of the complexities of our physical world and the remarkable properties of the most abundant substance on Earth. Further study of this fascinating anomaly continues to inspire scientists and reveal new insights into the fundamental workings of nature. The ability of ice to float is not just a curious scientific fact; it's a fundamental element supporting the complex web of life on our planet.
Latest Posts
Latest Posts
-
Dna Strands Are Antiparallel What Does That Mean
Apr 04, 2025
-
What Color In The Visible Spectrum Has The Longest Wavelength
Apr 04, 2025
-
Carbohydrates Are Used In Our Bodies Mainly For
Apr 04, 2025
-
Tap Water Mixture Or Pure Substance
Apr 04, 2025
-
Is Work A Scalar Or A Vector Quantity
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Ice Has Lower Density Than Water Because Ice . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
