How Many Molecules Are In 1 Mole Of Water
News Leon
Apr 05, 2025 · 6 min read
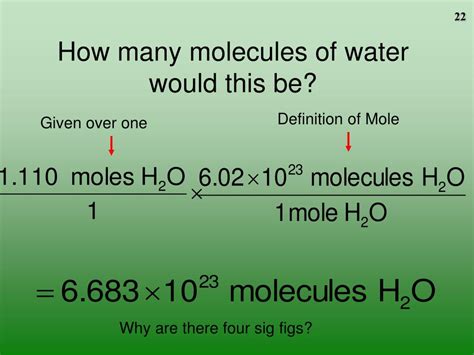
Table of Contents
How Many Molecules Are in 1 Mole of Water? A Deep Dive into Avogadro's Number
Understanding the relationship between moles, molecules, and Avogadro's number is fundamental to chemistry. This article will delve deep into this crucial concept, explaining not only how many molecules are in one mole of water but also the underlying principles and their broader implications.
What is a Mole?
Before we tackle the central question, let's clarify the concept of a mole. A mole (mol) is not a furry creature; instead, it's a fundamental unit in chemistry that represents a specific number of particles. This number, known as Avogadro's number, is approximately 6.022 x 10<sup>23</sup>. Therefore, one mole of any substance contains 6.022 x 10<sup>23</sup> particles of that substance. These particles can be atoms, molecules, ions, or even electrons, depending on the context.
Think of a mole like a dozen. A dozen eggs always contains 12 eggs, regardless of the size or type of eggs. Similarly, a mole of water always contains 6.022 x 10<sup>23</sup> water molecules.
The Importance of the Mole
The mole is incredibly important in chemistry because it allows us to bridge the gap between the microscopic world of atoms and molecules and the macroscopic world of laboratory measurements. We can't directly count individual atoms or molecules, but we can easily measure the mass of a substance. The mole provides a convenient way to relate mass to the number of particles.
This is achieved through the molar mass of a substance. The molar mass is the mass of one mole of that substance, usually expressed in grams per mole (g/mol). For example, the molar mass of water (H₂O) is approximately 18 g/mol. This means that one mole of water weighs 18 grams.
Calculating the Number of Molecules in 1 Mole of Water
Now, we can directly answer the main question: How many molecules are in 1 mole of water?
The answer, as stated earlier, is 6.022 x 10<sup>23</sup> molecules. This is because, by definition, one mole of any substance contains Avogadro's number of particles. In the case of water, the particles are water molecules (H₂O).
Understanding Avogadro's Number
Avogadro's number is not an arbitrary number; it's a carefully determined constant that relates the atomic mass unit (amu) to the gram. One amu is approximately the mass of a single proton or neutron. Avogadro's number ensures that the molar mass of a substance in grams is numerically equal to the average mass of one molecule or atom in atomic mass units.
This is a powerful concept because it simplifies calculations significantly. We can easily convert between grams, moles, and the number of molecules using Avogadro's number and molar mass.
Beyond Water: Applying the Concept to Other Substances
The principle of one mole containing 6.022 x 10<sup>23</sup> particles applies to all substances, not just water. Let's consider some examples:
- One mole of oxygen gas (O₂): Contains 6.022 x 10<sup>23</sup> oxygen molecules. Each molecule consists of two oxygen atoms.
- One mole of sodium chloride (NaCl): Contains 6.022 x 10<sup>23</sup> formula units of NaCl. In this case, we use the term "formula unit" instead of "molecule" because NaCl is an ionic compound, not a molecule.
- One mole of iron (Fe): Contains 6.022 x 10<sup>23</sup> iron atoms.
The key is to understand that the mole is a counting unit, and Avogadro's number is the count itself, irrespective of the chemical nature of the substance.
Practical Applications and Significance
The mole concept is not merely an abstract idea; it's crucial for numerous practical applications in chemistry and related fields:
- Stoichiometry: The mole allows us to perform stoichiometric calculations, which are essential for determining the amounts of reactants and products in chemical reactions.
- Titrations: Titrations rely heavily on the mole concept to determine the concentration of unknown solutions.
- Chemical Synthesis: In chemical synthesis, the mole is used to precisely control the amounts of reactants to achieve desired yields.
- Environmental Science: The mole is used to measure pollutant concentrations and assess environmental impacts.
- Materials Science: In materials science, understanding the number of atoms and molecules in a material is crucial for controlling its properties.
Further Exploring Avogadro's Number and its History
Avogadro's number is named after Amedeo Avogadro, an Italian scientist who, in 1811, proposed that equal volumes of gases at the same temperature and pressure contain equal numbers of molecules. However, it wasn't until much later that Avogadro's number was accurately determined through various experimental techniques. These techniques often involved measuring the charge of a single electron and using this to estimate the number of particles in a known amount of substance.
The precise determination of Avogadro's number continues to be a subject of ongoing research. Sophisticated techniques like X-ray crystallography are used to refine its value and contribute to a deeper understanding of fundamental physical constants.
Molar Mass and its Calculation
Understanding molar mass is crucial for using the mole concept effectively. The molar mass of a substance is the mass of one mole of that substance in grams. It's calculated by adding the atomic masses of all the atoms in the chemical formula of the substance.
Let's consider water (H₂O) again:
- Atomic mass of Hydrogen (H) ≈ 1.01 g/mol
- Atomic mass of Oxygen (O) ≈ 16.00 g/mol
Therefore, the molar mass of H₂O = (2 x 1.01 g/mol) + (1 x 16.00 g/mol) ≈ 18.02 g/mol
This means that one mole of water weighs approximately 18.02 grams.
Conversions Involving Moles, Mass, and Number of Molecules
We can use molar mass and Avogadro's number to seamlessly convert between grams, moles, and the number of molecules. Here's a breakdown of the conversion factors:
- Grams to moles: Divide the mass in grams by the molar mass (g/mol).
- Moles to grams: Multiply the number of moles by the molar mass (g/mol).
- Moles to number of molecules: Multiply the number of moles by Avogadro's number (6.022 x 10<sup>23</sup>).
- Number of molecules to moles: Divide the number of molecules by Avogadro's number (6.022 x 10<sup>23</sup>).
Addressing Common Misconceptions
One common misconception is that Avogadro's number is exact. It's not; it's an experimentally determined value with a degree of uncertainty. The value 6.022 x 10<sup>23</sup> is an approximation.
Another misconception is that the mole concept applies only to molecules. It applies to atoms, ions, formula units, and any other defined particle. The essential factor is that it represents Avogadro's number of entities.
Conclusion
Understanding the number of molecules in one mole of water (and any substance) is crucial for mastering fundamental chemical concepts. The mole, Avogadro's number, and molar mass are interconnected tools that allow chemists to bridge the gap between the microscopic and macroscopic worlds, making accurate quantitative calculations in chemistry possible. This understanding underpins countless applications in various scientific disciplines and technological advancements. This deep dive into the topic has hopefully clarified this important concept and highlighted its significance in various fields of study.
Latest Posts
Latest Posts
-
Which Of The Following Is An Assumption Of Theory Y
Apr 05, 2025
-
Write The Electronic Configuration Of Sodium
Apr 05, 2025
-
Is The Square Root Of 7 Irrational
Apr 05, 2025
-
Calculate The Mass Of Br 79
Apr 05, 2025
-
How Many Ounces Are In 2 Lbs
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Many Molecules Are In 1 Mole Of Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
